Please select a subject first
Advertisements
Advertisements
4 L of 0.02 M aqueous solution of NaCl was diluted by adding one litre of water. The molality of the resultant solution is ______.
Concept: undefined >> undefined
Cone. H2SO4 is 98% H2SO4 by mass has d = 1.84 g cm−3. Volume of acid required to make one litre of 0.1 M H2SO4 is:
Concept: undefined >> undefined
Advertisements
What is the mole fraction of solute in 1.00 m aqueous solutions?
Concept: undefined >> undefined
\[\begin{array}{cc}
\ce{CH3-CH-CH=CH2 + HBr -> A}\\
|\phantom{.....................}\\
\ce{CH3}\phantom{..................}
\end{array}\]
'A' is:
Concept: undefined >> undefined
The heating of phenyl methyl ether with HI produces:
Concept: undefined >> undefined
Which of the following gives a positive iodoform test?
Concept: undefined >> undefined
\[\ce{Phenol ->[Zn, dust] 'X' ->[CH3Cl][Anhy. AlCl3] 'Y' ->[Alkaline][KMnO4] 'Z'}\]
The product ‘Z’ is:
Concept: undefined >> undefined
Among the following sets of reactants which one produces anisole?
Concept: undefined >> undefined
IUPAC name of the compound is:
\[\begin{array}{cc}
\ce{CH3-CH-OCH3}\\
|\phantom{....}\\
\ce{CH3}\phantom{..}
\end{array}\]
Concept: undefined >> undefined
The correct acidic strength order of the following is:

(I)
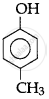
(II)

(III)
Concept: undefined >> undefined
Which of the following is a correct statement for C2H5Br?
Concept: undefined >> undefined
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Molarity of a solution changes with temperature.
Reason (R): Molarity is a colligative property.
Select the most appropriate answer from the options given below:
Concept: undefined >> undefined
Which of the following is not true about the ionic solids?
Concept: undefined >> undefined
An excess of potassium ions makes KCl crystals appear violet or lilac in colour since:
(i) some of the anionic sites are occupied by an unpaired electron.
(ii) some of the anionic sites are occupied by a pair of electrons.
(iii) there are vacancies at some anionic sites.
(iv) F-centres are created which impart colour to the crystals.
Concept: undefined >> undefined
Which of the following cannot be regarded as molecular solid?
(i) SiC (Silicon carbide)
(ii) AlN
(iii) Diamond
(iv) I2
Concept: undefined >> undefined
Why are liquids and gases categorised as fluids?
Concept: undefined >> undefined
Inspite of long range order in the arrangement of particles why are the crystals usually not perfect?
Concept: undefined >> undefined
Match the terms given in Column I with expressions given in Column II.
| Column I | Column II |
| (i) Mass percentage | (a) `"Number of moles of the solute component"/"Volume of solution in litres"` |
| (ii) Volume percentage | (b) `"Number of moles of a component"/"Total number of moles of all the components"` |
| (iii) Mole fraction | (c) `"Volume of the solute component in solution"/"Total volume of solution" xx 100` |
| (iv) Molality | (d) `"Mass of the solute component in solution"/"Total mass of the solution" xx 100` |
| (v) Molarity | (e) `"Number of moles of the solute components"/"Mass of solvent in kilograms"` |
Concept: undefined >> undefined
Define the following modes of expressing the concentration of a solution. Which of these modes are independent of temperature and why?
w/w (mass percentage)
Concept: undefined >> undefined
Define the following modes of expressing the concentration of a solution. Which of these modes are independent of temperature and why?
V/V (volume percentage)
Concept: undefined >> undefined
