Advertisements
Advertisements
Which of the following process is not responsible for the presence of electric charge on the sol particles?
Concept: undefined >> undefined
Which of the following phenomenon occurs when a chalk stick is dipped in ink?
(i) Adsorption of coloured substance.
(ii) Adsorption of solvent.
(iii) Absorption and adsorption both of solvent.
(iv) Absoprtion of solvent.
Concept: undefined >> undefined
Advertisements
Which of the following statements are correct?
(i) A depressant prevents certain type of particle to come to the froth.
(ii) Copper matte contains \[\ce{Cu2S}\] and \[\ce{ZnS}\].
(iii) The solidified copper obtained from reverberatory furnace has blistered appearance due to evolution of \[\ce{SO2}\] during the extraction.
(iv) Zinc can be extracted by self-reduction.
Concept: undefined >> undefined
What is the role of flux in metallurgical processes?
Concept: undefined >> undefined
Identify the compound Y in the following reaction.

Concept: undefined >> undefined
In which of the following molecules carbon atom marked with asterisk (*) is asymmetric?
(a) \[\begin{array}{cc}
\ce{H}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{.}\ce{I}\phantom{...}\ce{Br}\phantom{..}\ce{Cl}
\end{array}\]
(b) \[\begin{array}{cc}
\ce{D}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{.}\ce{I}\phantom{...}\ce{Br}\phantom{..}\ce{Cl}
\end{array}\]
(c) \[\begin{array}{cc}
\ce{H}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{..}\ce{OH}\phantom{..}\ce{C2H5}\phantom{}\ce{CH3}
\end{array}\]
(d) \[\begin{array}{cc}
\ce{H}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{..}\ce{H}\phantom{...}\ce{C2H5}\phantom{}\ce{CH3}
\end{array}\]
Concept: undefined >> undefined
Which of the following structures is enantiomeric with the molecule (A) given below:
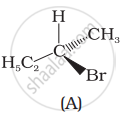
Concept: undefined >> undefined
Which of the following is an example of vic-dihalide?
Concept: undefined >> undefined
The position of \[\ce{-Br}\] in the compound in \[\ce{CH3CH = CH(Br)(CH3)2}\] can be classified as ______.
Concept: undefined >> undefined
Ethylidene chloride is a/an ______.
Concept: undefined >> undefined
Which is the correct IUPAC name for \[\begin{array}{cc}
\ce{CH3 - CH - CH2 - Br}\\
\phantom{}|\phantom{.......}\\
\phantom{}\ce{C2H5}\phantom{....}
\end{array}\]?
Concept: undefined >> undefined
What should be the correct IUPAC name for diethylbromomethane?
Concept: undefined >> undefined
The reaction of toluene with chlorine in the presence of iron and in the absence of light yields ______.
Concept: undefined >> undefined
Which of the carbon atoms present in the molecule given below are asymmetric?
\[\begin{array}{cc}
\phantom{}\ce{HO}\phantom{.....}\ce{OH}\phantom{..}\ce{H}\phantom{.....}\ce{O}\phantom{..}\\
\phantom{..}\backslash\phantom{.....}|\phantom{....}|\phantom{.....}//\phantom{.}\\
\ce{\overset{a}{C} - \overset{b}{C} - \overset{c}{C} - \overset{d}{C}}\\
\phantom{..}//\phantom{.....}|\phantom{....}|\phantom{....}\phantom{.}\backslash\phantom{...}\\
\phantom{}\ce{O}\phantom{......}\ce{H}\phantom{...}\ce{OH}\phantom{...}\ce{H}\phantom{}\\
\end{array}\]
Concept: undefined >> undefined
Haloalkanes contain halogen atom (s) attached to the sp3 hybridised carbon atom of an alkyl group. Identify haloalkane from the following compounds.
(i) 2-Bromopentane
(ii) Vinyl chloride (chloroethene)
(iii) 2-chloroacetophenone
(iv) Trichloromethane
Concept: undefined >> undefined
Which of the following compounds can be classified as aryl halides?
(i) \[\ce{p-ClC6H4CH2CH(CH3)2}\]
(ii) \[\ce{p-CH3CHCl(C6H4)CH2CH3}\]
(iii) \[\ce{o-BrH2C - C6H4CH(CH3)CH2CH3}\]
(iv) \[\ce{C6H5 - Cl}\]
Concept: undefined >> undefined
Which of the products will be major product in the reaction given below? Explain.
\[\ce{CH3CH = CH2 + HI -> \underset{(A)}{CH3CH2CH2I} + \underset{(B)}{CH3CHICH3}}\]
Concept: undefined >> undefined
Classify the following compound as a primary, secondary and tertiary halide.
2-Bromo-2-methylpropane
Concept: undefined >> undefined
Why can aryl halides not be prepared by reaction of phenol with \[\ce{HCl}\] in the presence of \[\ce{ZnCl2}\]?
Concept: undefined >> undefined
Match the items of Column I and Column II.
| Column I | Column II | |
| (i) | SN1 reaction | (a) vic-dibromides |
| (ii) | Chemicals in fire extinguisher | (b) gem-dihalides |
| (iii) | Bromination of alkenes | (c) Racemisation |
| (iv) | Alkylidene halides | (d) Saytzeff rule |
| (v) | Elimination of HX from alkylhalide | (e) Chlorobromocarbons |
Concept: undefined >> undefined
