Advertisements
Advertisements
प्रश्न
In \[\ce{PCl5}\], phosphorus is in sp3d hybridised state but all its five bonds are not equivalent. Justify your answer with reason.
टिप्पणी लिखिए
Advertisements
उत्तर
In \[\ce{PCl5}\], phosphorus undergoes sp3d hybridisation and a trigonal bipyramidal configuration comes into existence.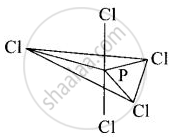
The equatorial \[\ce{P - Cl}\] bonds are equivalent while two axial bonds are different and larger than equatorial bonds.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
