Please select a subject first
Advertisements
Advertisements
Among the elements \[\ce{B, Al, C}\] and \[\ce{Si}\], which element has the most metallic character?
Concept: undefined >> undefined
Arrange the elements \[\ce{N, P, O}\] and \[\ce{S}\] in the order of increasing non metallic character. Give reason for the arrangement assigned.
Concept: undefined >> undefined
Advertisements
Among alkali metals which element do you expect to be least electronegative and why?
Concept: undefined >> undefined
What is meant by the term average bond enthalpy? Why is there difference in bond enthalpy of \[\ce{O - H}\] bond in ethanol \[\ce{(C2H5OH)}\] and water?
Concept: undefined >> undefined
The variation of pressure with volume of the gas at different temperatures can be graphically represented as shown in figure.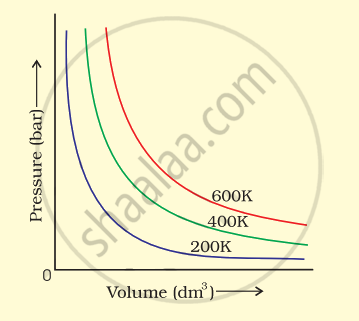
On the basis of this graph answer the following questions.
(i) How will the volume of a gas change if its pressure is increased at constant temperature?
(ii) At a constant pressure, how will the volume of a gas change if the temperature is increased from 200 K to 400 K?
Concept: undefined >> undefined
Match the following gas laws with the equation representing them.
| (i) Boyle's law | (a) `V oo n` at constant T and p |
| (ii) Charle's law | (b) PTotal = p1 + p2 + p3 + at constant T, V |
| (iii) Dalton's law | (c) `(pV)/T` = Constant T |
| (iv) Avogadro law | (d) `V oo T` at constant n and p |
| (e) `P oo 1/V` at constant n and T |
Concept: undefined >> undefined
The state of a gas can be described by quoting the relationship between ______.
Concept: undefined >> undefined
We know that the relationship between Kc and Kp is Kp = Kc (RT)∆n What would be the value of ∆n for the reaction \[\ce{NH4Cl (s) ⇌ NH3 (g) + HCl (g)}\]
Concept: undefined >> undefined
Why do beryllium and magnesium not impart colour to the flame in the flame test?
Concept: undefined >> undefined
What is the correct order of decreasing stability of the following cations.
| I. | \[\ce{CH3 - \overset{⊕}{C}H - CH3}\] |
| II. | \[\ce{CH3 - \overset{⊕}{C}H - OCH3}\] |
| III. | \[\ce{CH3 - \overset{⊕}{C}H - CH2 - OCH3}\] |
Concept: undefined >> undefined
Covalent bond can undergo fission in two different ways. The correct representation involving a heterolytic fission of CH3 – Br is:
Concept: undefined >> undefined
Write structures of various carbocations that can be obtained from 2-methylbutane. Arrange these carbocations in order of increasing stability.
Concept: undefined >> undefined
Match the intermediates given in Column I with their probable structure in Column II.
| Column I | Column II |
| (i) Free radical | (a) Trigonal planar |
| (ii) Carbocation | (b) Pyramidal |
| (iii) Carbanion | (c) Linear |
Concept: undefined >> undefined
Which of the following practices will not come under green chemistry?
Concept: undefined >> undefined
An alkene ‘A’ contains three C – C, eight C – H σ bonds and one C – C π bond. ‘A’ on ozonolysis gives two moles of an aldehyde of molar mass 44 u. Write IUPAC name of ‘A’.
Concept: undefined >> undefined
An alkene ‘A’ contains three C–C, eight C–H σ bonds and one C–C π bond. ‘A’ on ozonolysis gives two moles of an aldehyde of molar mass 44 u. Write IUPAC name of ‘A’.
Concept: undefined >> undefined
The following data are obtained when dinitrogen and dioxygen react together to form different compounds:-
| Mass of dinitrogen | Mass of dioxygen | |
| (i) | 14 g | 16 g |
| (ii) | 14 g | 32 g |
| (iii) | 28 g | 32 g |
| (iv) | 28 g | 80 g |
(a) Which law of chemical combination is obeyed by the above experimental data?Give its statement.
(b) Fill in the blanks in the following conversions:
(i) 1 km = ______ mm = ______ pm
(ii) 1 mg = ______ kg = ______ ng
(iii) 1 mL = ______ L = _______ dm3
Concept: undefined >> undefined
How do you express the bond strength in terms of bond order?
Concept: undefined >> undefined
Compare the relative stability of the following species and indicate their magnetic properties;
`"O"_2, "O"_2^+, "O"_2^-`(superoxide), `"O"_2^(2-)`(peroxide)
Concept: undefined >> undefined
Calculate the bond order of N2, O2, `"O"_2^+`and `"O"_2^-`?
Concept: undefined >> undefined
