Advertisements
Advertisements
प्रश्न
What is meant by the term average bond enthalpy? Why is there difference in bond enthalpy of \[\ce{O - H}\] bond in ethanol \[\ce{(C2H5OH)}\] and water?
Advertisements
उत्तर
The average bond enthalpy is defined as the ratio of total bond dissociation enthalpy to the number of bonds broken in the structure.
The identical \[\ce{O - H}\] bonds in water molecule does not have the same bond enthalpies. According to the structure of a water molecule, there are two \[\ce{O - H}\] bonds, but there is a change in the breaking of the first \[\ce{O - H}\] bond than the second because of the different charge.
Hence in water molecule the average bond enthalpy will be:
The average \[\ce{O - H}\] bond enthalpy = (502 + 427)/2 = 464 kJ mol–1.
In ethanol \[\ce{C2H5OH}\] the bond enthalpy of \[\ce{O - H}\] is different because the electronic environment around oxygen atom is different. In ethanol the \[\ce{O - H}\] is attached to the carbon atom and in water molecule the \[\ce{O - H}\] is attached to the hydrogen atom.
APPEARS IN
संबंधित प्रश्न
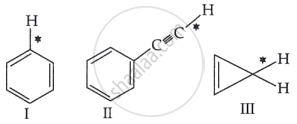
Among the given compounds I-III, the correct order of bond dissociation energy of the C-H bond marked with * is:
Identify the correct orders against the property mentioned:
A. H2O > NH3 > CHCl3 - dipole moment
B. XeF4 > XeO3 > XeF2 - number of lone pairs on central atom
C. O-H > C-H > N-O - bond length
D. N2 > O2 > H2 - bond enthalpy
Choose the correct answer from the options given below:
