Please select a subject first
Advertisements
Advertisements
What do you understand by isoelectronic species? Name a species that will be isoelectronic with the following atom or ion:-
F-
Concept: undefined >> undefined
What do you understand by isoelectronic species? Name a species that will be isoelectronic with the following atom or ion:-
Ar
Concept: undefined >> undefined
Advertisements
What do you understand by isoelectronic species? Name a species that will be isoelectronic with the following atom or ion:-
Mg2+
Concept: undefined >> undefined
What do you understand by isoelectronic species? Name a species that will be isoelectronic with the following atom or ion:-
Rb+
Concept: undefined >> undefined
Consider the following species:-
N3–, O2–, F–, Na+, Mg2+ and Al3+
Arrange them in the order of increasing ionic radii.
Concept: undefined >> undefined
Consider the following species:-
N3–, O2–, F–, Na+, Mg2+ and Al3+
What is common in them?
Concept: undefined >> undefined
Explain why cations are smaller and anions larger in radii than their parent atoms?
Concept: undefined >> undefined
The size of isoelectronic species — F-, Ne and Na+ is affected by ______.
Concept: undefined >> undefined
Explain the important aspects of resonance with reference to the `"CO"_3^(2-)` ion.
Concept: undefined >> undefined
H3PO3 can be represented by structures 1 and 2 shown below. Can these two structures be taken as the canonical forms of the resonance hybrid representing H3PO3? If not, give reasons for the same.
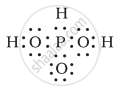 |
 |
| (1) | (2) |
Concept: undefined >> undefined
Write the resonance structure for SO3.
Concept: undefined >> undefined
Write the resonance structures for NO2.
Concept: undefined >> undefined
Write the resonance structures for `"NO"_3^(-)`.
Concept: undefined >> undefined
At 0°C, the density of a certain oxide of a gas at 2 bar is same as that of dinitrogen at 5 bar. What is the molecular mass of the oxide?
Concept: undefined >> undefined
The pressure of 1 g of an ideal gas A at 27 °C is found to be 2 bar. When 2 g of another ideal gas B is introduced in the same flask at the same temperature the pressure becomes 3 bar. Find a relationship between their molecular masses.
Concept: undefined >> undefined
The drain cleaner, Drainex contains small bits of aluminum which react with caustic soda to produce dihydrogen. What volume of dihydrogen at 20 °C and one bar will be released when 0.15g of aluminum reacts?
Concept: undefined >> undefined
The density of a gas is found to be 5.46 g/dm3 at 27 °C at 2 bar pressure. What will be its density at STP?
Concept: undefined >> undefined
How much time would it take to distribute one Avogadro number of wheat grains, if 1010 grains are distributed each second?
Concept: undefined >> undefined
Comment on the thermodynamic stability of NO(g), given
\[\ce{1/2 N_2 (g) + 1/2 O2(g) → NO(g)}\]; ΔrHθ = 90 kJ mol–1
\[\ce{NO(g) +1/2 O2(g) → NO2(g)}\] : ΔrHθ= –74 kJ mol–1
Concept: undefined >> undefined
Why is Wurtz reaction not preferred for the preparation of alkanes containing odd number of carbon atoms? Illustrate your answer by taking one example
Concept: undefined >> undefined
