Please select a subject first
Advertisements
Advertisements
A food chain comprising birds, green plants, fish and man. The concentration of harmful chemical entering the food chain will be maximum in ______.
Concept: undefined >> undefined
Which of the following is a logical sequence of food chain?
Concept: undefined >> undefined
Advertisements
Which of the following is an autotroph?
Concept: undefined >> undefined
Magnetic lines of force inside current carrying solenoid are ____________.
Concept: undefined >> undefined
Food chains are very important for the survival of most species.

If 10,000 J solar energy falls on green plants in a terrestrial ecosystem, what percentage of solar energy will be converted into food energy?
Concept: undefined >> undefined
Food chains are very important for the survival of most species.

Matter and energy are two fundamental inputs of an ecosystem. Movement of:
Concept: undefined >> undefined
Observe the food web and answer the questions given below:
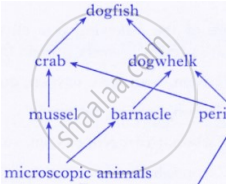
Why do all food chains start with plants?
Concept: undefined >> undefined
Which of the following is not a physical change?
Concept: undefined >> undefined
The following reaction is an example of a `4"NH"_3("g") + "SO"_2 -> 4"NO"("g") + 6"H"_2"O"("g")`
- displacement reaction
- combination reaction
- redox reaction
- neutralisation reaction
Concept: undefined >> undefined
Which of the following statements about the given reaction are correct?
`3"Fe"("s") + 4"H"_2"O"("g") -> "Fe"_3"O"_4("s") + 4"H"_2("g")`
- Iron metal is getting oxidised
- Water is getting reduced
- Water is acting as reducing agent
- Water is acting as oxidising agent
Concept: undefined >> undefined
Which of the following are exothermic processes?
- Reaction of water with quick lime
- Dilution of an acid
- Evaporation of water
- Sublimation of camphor (crystals)
Concept: undefined >> undefined
Three beakers labelled as A, B and C each containing 25 mL of water were taken. A small amount of NaOH, anhydrous CuSO4 and NaCl were added to the beakers A, B and C respectively. It was observed that there was an increase in the temperature of the solutions contained in beakers A and B, whereas in case of beaker C, the temperature of the solution falls. Which one of the following statement(s) is(are) correct?
- In beakers A and B, exothermic process has occurred.
- In beakers A and B, endothermic process has occurred.
- In beaker C exothermic process has occurred.
- In beaker C endothermic process has occurred.
Concept: undefined >> undefined
A dilute ferrous sulphate solution is gradually added to the beaker containing acidified permanganate solution. The light purple colour of the solution fades and finally disappears. Which of the following is the correct explanation for the observation?
Concept: undefined >> undefined
Which among the following is(are) double displacement reaction(s)?
- `"Pb" + "CuCl"_2 -> "PbCl"_2 + "Cu"`
- `"Na"_2"SO"_4 + "BaCl"_2 -> "BaSO"_4 + 2"NaCl"`
- `"C" + "O"_2 -> "CO"_2`
- `"CH"_4 + 2"O"_2 -> "CO"_2 + 2"H"_2"O"`
Concept: undefined >> undefined
Which among the following statement(s) is(are) true? ‘Exposure of silver chloride to sunlight for a long duration turns grey due to
- The formation of silver by the decomposition of silver chloride.
- sublimation of silver chloride.
- decomposition of chlorine gas from silver chloride.
- oxidation of silver chloride.
Concept: undefined >> undefined
Give the characteristic tests for the following gases :
- CO2
- SO2
- O2
- H2
Concept: undefined >> undefined
An element A is soft and can be cut with a knife. This is very reactive to air and cannot be kept open in air. It reacts vigorously with water. Identify the element from the following
Concept: undefined >> undefined
Although metals form basic oxides, which of the following metals form an amphoteric oxide?
Concept: undefined >> undefined
A molecule of ammonia (NH3) has
Concept: undefined >> undefined
Which of the following are correct structural isomers of butane?
- \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - C - H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
\end{array}\] - \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - H}\\
|\phantom{.....}|\phantom{.....}|\\
\ce{H}\ce{H-C-H}\ce{H}\\
|\\
\ce{H}\\
\end{array}\] - \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - H}\\
|\phantom{.....}\backslash\phantom{..}|\\
\phantom{....}\ce{H}\phantom{......}\ce{C - H}\phantom{}\\
\phantom{.......}|\\
\phantom{.......}\ce{H}\\
\end{array}\] - \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\\
\ce{H - C - C - H}\\
|\phantom{....}|\\
\ce{H - C - C - H}\\
|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}\\
\end{array}\]
Concept: undefined >> undefined
