Advertisements
Advertisements
The complex ion `[Co(H_2O)_5 (ONO)]^(2+) `
(A) linkage isomer
(B) ionisation isomer
(C) co-ordination isomer
(D) geometrical isomer
Concept: Isomerism in Coordination Compounds >> Structural Isomerism
Write the structural formula and IUPAC names of all possible isomers of the compound with molecular formula C3H8O.
Concept: Isomerism in Coordination Compounds
Calculate the effective atomic number of the central metal atom in the following compounds:
K4Fe(CN)6 Fe(Z=26)
Concept: Concept of Coordination Compounds >> Importance of Coordination Compounds
Calculate the effective atomic number of the central metal atom in the following compounds:
K4Fe(CN)6 Fe(Z=26)
Concept: Concept of Coordination Compounds >> Importance of Coordination Compounds
Effective atomic number rule is used to find _______.
(A) geometry of complex
(B) stability of complex
(C) number of isomers of complex
(D) number of possible ligands around metal ion in complex
Concept: Concept of Coordination Compounds >> Importance of Coordination Compounds
Effective atomic number rule is used to find _______.
(A) geometry of complex
(B) stability of complex
(C) number of isomers of complex
(D) number of possible ligands around metal ion in complex
Concept: Concept of Coordination Compounds >> Importance of Coordination Compounds
How ligands are classified? Explain with suitable examples.
Concept: Concept of Coordination Compounds >> Introduction
Which of the following compounds is used as a semipermeable membrane?
(a) Potassium ferrocyanide
(b) Potassium argentocyanide
(c) Sodium meta aluminate
(d) Copper ferrocyanide
Concept: Important Terms Pertaining to Coordination Compounds
Predict the co-ordination No. of cs+ ion if `r_(Cs)^+` = 1.69Å and `r_(Cl)^-` = 1.81Å.
Concept: Important Terms Pertaining to Coordination Compounds
IUPAC name of the following compound is
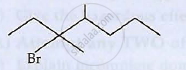
(a) 3 - Bromo- 3, 4- dimethylheptane
(b) 3, 4- dimethyl - 3- bromoheptane
(c) 5- Bromo- 4, 5- dimethylheptane
(d) 4, 5- dimethyl- 5- bromoheptane
Concept: Important Terms Pertaining to Coordination Compounds
Write IUPAC names of the following compounds

Concept: Important Terms Pertaining to Coordination Compounds
Write IUPAC names of the following compounds:

Concept: Important Terms Pertaining to Coordination Compounds
Write four postulates of Werner's theory.
Concept: Werner’s Theory of Coordination Compounds
How are the following conversions carried out?
Benzoic acid into metanitrobenzoic acid.
Concept: Bonding in Coordination Compounds >> Crystal Field Theory (CFT)
State the superiority of crystal field theory over valence bond theory.
Concept: Bonding in Coordination Compounds >> Crystal Field Theory (CFT)
Write applications of co-ordination compounds in medicine and electroplating.
Concept: Important Terms Pertaining to Coordination Compounds
Calculate EAN of cobalt (Z=27) in [CO(NH3)6]+3 and of zinc (Z=30) in [Zn(NH3)4]SO4.
Concept: Concept of Coordination Compounds >> Importance of Coordination Compounds
Calculate EAN of cobalt (Z=27) in [CO(NH3)6]+3 and of zinc (Z=30) in [Zn(NH3)4]SO4.
Concept: Concept of Coordination Compounds >> Importance of Coordination Compounds
Calculate the effective atomic number of the central metal atom in the following compounds:
Cr(CO)6 Cr(Z=24)
Concept: Concept of Coordination Compounds >> Importance of Coordination Compounds
Calculate the effective atomic number of the central metal atom in the following compounds:
Cr(CO)6 Cr(Z=24)
Concept: Concept of Coordination Compounds >> Importance of Coordination Compounds
