Please select a subject first
Advertisements
Advertisements
Do the following conversions in not more than two steps:
CH3COOH to CH3COCH3
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Structure of the Carbonyl Group
Do the following conversions in not more than two steps:
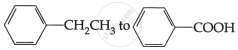
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Structure of the Carbonyl Group
Assertion (A): Acetic acid but not formic acid, can be halogenated in the presence of red P and Cl2.
Reason (R): Acetic acid is a weaker acid than formic acid.
Concept: Acids >> Chemical Reactions of Carboxylic Acids - Substitution Reactions in the Hydrocarbon Part
Assertion (A): The final product in Aldol condensation is always α, β-unsaturated carbonyl compound.
Reason (R): α, β-unsaturated carbonyl compounds are stabilised due to conjugation.
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen

Identify A and B:
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
What is IUPAC name of the ketone A, which undergoes iodo form reaction to give \[\ce{CH3CH = C(CH3)COONa}\] and yellow precipitate of \[\ce{CH3}\]?
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Nomenclature of Aldehydes and Ketones
Assertion (A): Strong oxidising agents oxidise toluene and its derivatives to benzoic acids.
Reason (R): It is possible to stop the oxidation of toluene at the aldehyde stage with suitable reagents.
Select the most appropriate answer from the options given below:
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Preparation of Aldehydes
Out of p-tolualdehyde and p-nitrobenzaldehyde, which one is more reactive towards nucleophilic addition reactions, why?
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
Write the structure of the product formed when acetone reacts with 2, 4 DNP reagent.
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
Convert the following:
Benzene to m-nitrobenzaldehyde
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Chemical Reactions of Aldehydes and Ketones - Other Reactions
Convert the following:
Bromobenzene to benzoic acid
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Nomenclature of Aldehydes and Ketones
You are given four organic compounds “A”, “B” , “C” and “D”. The compounds “A”, “B” and “C” form an orange-red precipitate with 2, 4 DNP reagent. Compounds “A” and “B” reduce Tollen’s reagent while compounds “C” and “D” do not. Both “B” and “C” give a yellow precipitate when heated with iodine in the presence of NaOH. Compound “D” gives brisk effervescence with sodium bicarbonate solution. Identify “A”, “B”, “C” and “D” given the number of carbon atoms in three of these carbon compounds is three while one has two carbon atoms. Give an explanation for our answer.
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Chemical Reactions of Aldehydes and Ketones - Oxidation
An organic compound with molecular formula \[\ce{C7H7NO2}\] exists in three isomeric forms, the isomer ‘A’ has the highest melting point of the three. ‘A’ on reduction gives compound ‘B’ with molecular formula \[\ce{C7H9N}\]. ‘B’ on treatment with \[\ce{NaNO2/HCl}\] at 0-5° C to form compound ‘C’. On treating C with \[\ce{H3PO2}\], it gets converted to D with formula \[\ce{C7H8}\], which on further reaction with \[\ce{CrO2Cl2}\] followed by hydrolysis forms ‘E’ \[\ce{C7H6O}\]. Write the structure of compounds A to E. Write the chemical equations involved.
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Preparation of Aldehydes
Account for the following:
N-ethylbenzene sulphonyl amide is soluble in alkali.
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Preparation of Aldehydes
Account for the following:
Reduction of nitrobenzene using Fe and HCl is preferred over Sn and HCl.
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Preparation of Aldehydes
Write the name of the reaction, structure and IUPAC name of the product formed when:
CH3CH2CN reacts with stannous chloride in the presence of hydrochloric acid, followed by hydrolysis.
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Preparation of Aldehydes
Give the structures of A, B and C in the following reactions:

Concept: Diazonium Salts >> Chemical Reaction of Diazonium Salts - Reactions Involving Displacement of Nitrogen
Write the major products(s) in the following:

Concept: Diazonium Salts >> Importance of Diazonium Salts in Synthesis of Aromatic Compounds
Distinguish between the following pairs of compounds: Aniline and N-methylaniline
Concept: Concept of Amines >> Identification of Primary, Secondary and Tertiary Amines
Write the structures of main products when benzene diazonium chloride `(C_6H_5N_2^(+)Cl^-)`reacts with the following reagents:
CuCN/KCN
Concept: Diazonium Salts >> Chemical Reaction of Diazonium Salts - Reactions Involving Displacement of Nitrogen
