- Ethyne (C₂H₂) is the first member of the alkyne series and is commonly known as acetylene.
- It has a linear structure with a triple bond between two carbon atoms.
- Sources include coal, gas, and petroleum cracking products.
- Electron Dot Formula:

- Structural Formula:
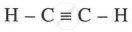
Definitions [1]
Definition: Alkynes
Alkynes are aliphatic unsaturated hydrocarbons containing at least one C≡C triple bond.
Key Points
Key Points: Alkynes
- General formula: CₙH₂ₙ₋₂
- Suffix for IUPAC naming: –yne
- e.g., Propyne (CH₃–C≡CH), Butyne (CH₃–CH₂–C≡CH)
- C₂H₂ is acetylene (common name); IUPAC name is ethyne
- The triple bond consists of one σ bond and two π bonds
Isomerism in Alkynes:
- Alkynes show position isomerism (type of structural isomerism)
- e.g., 1-Butyne and 2-Butyne
Key Points: Ethyne
Key Points: Physical Properties of Ethyne
- Ethyne is a colourless gas with a sweet smell when pure; impure gas may smell like garlic.
- Slightly soluble in water, but highly soluble in organic solvents like acetone and alcohol.
- The boiling point is –75°C, and it liquefies at –84°C; it is lighter than air (VD = 13).
Key Points: Chemical Properties of Ethyne
- Ethyne burns with a sooty flame due to high carbon content; in excess air, it gives CO₂, water, and heat.
- Undergoes addition reactions—adds hydrogen to form ethene and then ethane; adds halogens to form saturated compounds.
- Reacts with HCl to form chloroethene, which then undergoes stepwise addition to form 1,1-dichloroethane.
Key Points: Uses of Ethyne
