- Carboxylic acids are carbon compounds with a –COOH group and have acidic nature.
- Types:
- Monocarboxylic acids have one –COOH group (e.g., formic acid, acetic acid)
- Dicarboxylic acids have two –COOH groups (e.g., oxalic acid) - IUPAC Naming: Replace the ‘e’ of the corresponding alkane with ‘oic acid’ (e.g., ethane → ethanoic acid); also called alkanoic acids.
Definitions [3]
Definition: Carbonyl Compound
Organic compounds containing carbon-oxygen double bond, i.e. \[\mathrm{>C=O}\] group, are known as carbonyl compounds.
Definition: Carbonyl Compound
Organic compounds containing carbon-oxygen double bond, i.e. \[\mathrm{>C=O}\] group, are known as carbonyl compounds.
Definition: Carboxylic Acid
An organic compound containing the carboxyl group (-COOH) is known as carboxylic acid. These compounds possess acidic properties.
Formulae [1]
Formula: Carboxylic Acid
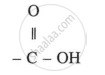
General formula: CnH2n+1COOH (or RCOOH)
Functional group:
Key Points
Key Points: Concepts of Aldehydes, Ketones, and Carboxylic Acids
- Carbonyl group: The ≻C=O group (carbonyl carbon + carbonyl oxygen) — a key functional group in organic chemistry.
- Carbonyl compounds: Aldehydes and ketones, both containing ≻C=O as their functional group.
- Aldehydes: –CHO (formyl group); carbonyl C bonded to at least one H.
- Ketones: ≻C=O (ketonic carbonyl group); carbonyl C bonded to two alkyl/aryl groups (R=R′ or R≠R′).
- Carboxylic acids: –COOH (carboxyl group); –OH attached to ≻C=O makes them distinct from aldehydes/ketones.
Key Points: Concepts of Aldehydes, Ketones, and Carboxylic Acids
- Carbonyl group: The ≻C=O group (carbonyl carbon + carbonyl oxygen) — a key functional group in organic chemistry.
- Carbonyl compounds: Aldehydes and ketones, both containing ≻C=O as their functional group.
- Aldehydes: –CHO (formyl group); carbonyl C bonded to at least one H.
- Ketones: ≻C=O (ketonic carbonyl group); carbonyl C bonded to two alkyl/aryl groups (R=R′ or R≠R′).
- Carboxylic acids: –COOH (carboxyl group); –OH attached to ≻C=O makes them distinct from aldehydes/ketones.
Key Points: Preparation of Aldehydes and Ketones
- IUPAC rules: Longest chain with –CHO/–COOH is parent; –CHO/–COOH carbon gets number 1; two ≻C=O in ketones → suffix "dione".
- Preparation (common methods): Oxidation of 1° alcohols → aldehyde, 2° alcohols → ketone; dehydrogenation (Cu, 575 K); ozonolysis of alkenes; hydration of alkynes (dil. H₂SO₄ + HgSO₄, 333 K).
- Aldehyde-specific preparations: Rosenmund reduction (RCOCl, H₂/Pd–BaSO₄); Stephen reaction (RCN, SnCl₂·HCl, H₃O⁺); DIBAL-H on nitriles/esters; Etard reaction, Gatterman–Koch formylation for aromatic aldehydes.
- Ketone-specific preparations: Acyl chloride + R₂Cd; nitrile + R′MgX/dry ether, H₃O⁺; Friedel–Crafts acylation of benzene with RCOCl/anhyd. AlCl₃.
- Key reactions: HCN → cyanohydrin; NaHSO₃ → bisulphite adduct; R′OH/HCl → acetal/ketal; RMgX → 2°/3° alcohol; NH₂OH → oxime; NH₂NH₂ → hydrazone; Clemmensen (Zn-Hg/HCl) & Wolf–Kishner (NH₂NH₂/KOH) → alkane; K₂Cr₂O₇/H₂SO₄ → carboxylic acid.
Key Points: Physical Properties of Aldehydes and Ketones
- Preparation (aliphatic/aromatic): From 2° alcohol (K₂Cr₂O₇/H₂SO₄ or Cu/573 K), alkene (ozonolysis), alkyne (dil. H₂SO₄/HgSO₄, 333 K), acetyl chloride + R₂Cd, nitrile + R′MgX (dry ether, H₃O⁺).
- Aromatic ketones: Benzene + RCOCl / anhyd. AlCl₃ (Friedel–Crafts acylation).
- Addition reactions: HCN → cyanohydrin; NaHSO₃ → bisulphite adduct; R″OH/dry HCl → ketal; R′MgX then H⁺/H₂O → 3° alcohol.
- Condensation reactions: NH₂OH → ketoxime; NH₂NH₂ → hydrazone; C₆H₅NHNH₂ → phenylhydrazone.
- Reductions: Clemmensen (Zn–Hg/conc. HCl) and Wolf–Kishner (NH₂NH₂, KOH/ethylene glycol) → alkane (R–CH₂–R′).
Key Points: Carboxylic Acid
Key Points: Physical Properties of Carboxylic Acids
- Solubility: Decreases with an increase in the size of the hydrocarbon part.
- Miscibility: Lower carboxylic acids (up to 4 C atoms) are miscible with water due to H-bonding.
- Boiling point: Carboxylic acids have higher B.P. than ketones, aldehydes, and alcohols of comparable molecular mass due to intermolecular H-bonding.
- Order of B.P. (carboxylic acids & aldehydes): Valeric > Butyric > Propionic > Acetic > Formic acid; Hexanal > Pentanal > Butanal > Propanal.
- Order of B.P. (ketones): Hexan-2-one > Pentan-2-one > Butan-2-one > Propanone.
Concepts [25]
- Concepts of Aldehydes, Ketones, and Carboxylic Acids
- Concepts of Aldehydes, Ketones, and Carboxylic Acids
- Nomenclature of Aldehydes and Ketones
- Nature of Carbonyl Group
- Structure of the Carbonyl Group
- Preparation of Aldehydes and Ketones
- Preparation of Aldehydes
- Preparation of Ketones
- Physical Properties of Aldehydes and Ketones
- Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
- Chemical Reactions of Aldehydes and Ketones - Reduction
- Chemical Reactions of Aldehydes and Ketones - Oxidation
- Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
- Chemical Reactions of Aldehydes and Ketones - Other Reactions
- Uses of Aldehydes and Ketones
- Carboxylic Acids
- Nomenclature of Carboxylic Acids
- Structure of the Carboxyl group
- Methods of Preparation of Carboxylic Acids
- Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of O-H Bond
- Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of C-OH Bond
- Chemical Reactions of Carboxylic Acids - Reactions Involving –COOH Group
- Chemical Reactions of Carboxylic Acids - Substitution Reactions in the Hydrocarbon Part
- Physical Properties of Carboxylic Acids
- Uses of Carboxylic Acids
