English Medium
Academic Year: 2025-2026
Date & Time: 25th February 2026, 10:30 am
Duration: 3h
Advertisements
General Instructions:
Read the following instructions carefully and follow them:
- This question paper contains 39 questions. All questions are compulsory.
- Question paper is divided into THREE sections - A, B and C.
- SECTION A: Biology (30 marks)
- SECTION B: Chemistry (25 marks)
- SECTION C: Physics (25 marks)
- The question paper has MCQs, VSAS, SAS, LAs and CBQs. Marks are given against each question.
- There are Cases Based Questions (CBQs) with three sub-questions and are of 4 marks each.
- Divide your answer sheet into three sections as per question paper - SECTION A (Biology), SECTION B (Chemistry) and SECTION C (Physics). It is compulsory to answer each question in its respective section. Do not mix answers of one section into the other section.
- Instructions are given with each section and question, wherever necessary.
- Kindly note that a separate question paper has been provided for visually impaired candidates.
- There is no overall choice in the question paper. However, an internal choice has been provided in few questions. Only one of the choices in such must be attempted.
Identify the part that controls the closing and opening of the stomatal pore in leaves of plants.
Select the correct option.
Stomata
Epidermal cells
Guard cells
Chloroplasts
Chapter:
Identify the endocrine gland that regulates the growth of human body.
Pituitary gland
Thyroid gland
Pancreas
Adrenal gland
Chapter:
Choose the equation of reaction that correctly represents anaerobic respiration in muscles:
\[\ce{Glucose -> Pyruvate ->[Absence][of O2] Ethanol + CO2 + Energy}\]
\[\ce{Glucose ->[O2] Pyruvate ->[O2] Energy}\]
\[\ce{Glucose ->[O2] Pyruvate ->[Lack of][oxygen] Ethanol + Energy}\]
\[\ce{Glucose -> Pyruvate ->[Lack of][oxygen] Lactic acid + Energy}\]
Chapter:
In human beings, the implantation of fertilised egg takes place in which part of female reproductive system?
Oviduct
Cervix
Uterus
Vagina
Chapter:
When a human egg is fertilized by a sperm having ‘Y’ chromosome, the zygote has the following combination of chromosomes:
44 + XX
22 + XX
44 + XY
22 + XY
Chapter:
Which of the following is a non-biodegradable pollutant?
Paper
DDT
Wood
Vegetable peel
Chapter:
In an ecosystem, the decomposers perform an important role. Select the option indicating the role of decomposers.
Convert inorganic materials to simpler forms
Break down complex organic substances to simple inorganic substances.
Break down inorganic substances into organic substances.
Do not act on organic compounds.
Chapter:
Assertion (A): Bacteria that can withstand heat have better chances of survival in a heat wave.
Reason (R): Accumulation of variations in a species increases the chances of its survival in changing environment.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (А).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
Assertion (A): The peristaltic movements occur all along the gut.
Reason (R): The lining of the gut has muscles that contract rhythmically to push the food in regulated manner.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
Give a diagrammatic representation of nutrition in Amoeba.
Chapter:
Observe the given figure and identify the labelled parts P, Q, R and S:
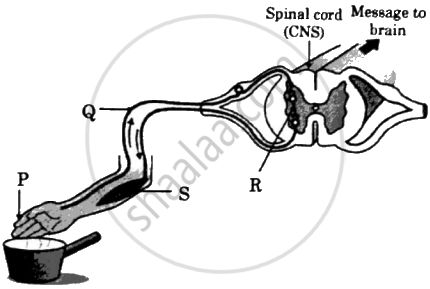
Chapter:
Which of the plant hormone is responsible for the following process?
Promote cell division
Chapter:
Which of the plant hormone is responsible for the following process?
Inhibition of growth
Chapter:
Which of the plant hormone is responsible for the following process?
Detection of light
Chapter:
Which of the plant hormones is responsible for the following process?
Wilting of leaves
Chapter:
Briefly mention the steps in double-circulation through human heart.
Chapter:
Based on Mendel’s experiment show the cross between round, yellow seeds of pea plant (RRYY) with wrinkled and green seeds of pea plant (rryy) upto F2 generation. Write the number of seeds/plants with following genetic (Genotype) makeup in F2 generation:
- RRYY
- rryy
Chapter:
Mention any one harmful effect of using plastic bags on the environment. Suggest better alternatives to the usage of plastic bags.
Chapter:
Paddy fields require a large amount of water. The pesticides and chemical fertilizers used are washed down into the soil or waterbodies. How do these chemicals reach our bodies? What is this phenomenon known as?
Chapter:
|
Kidneys help to regulate the volume of fluid and various metabolites waste products in the body. Normally, kidney filter about 180 L of fluid daily but the volume actually excreted out is only a litre or two a day. Patients with kidney failures can be saved by dialysis and kidney transplant. |
(a) Write the structure and function of Bowman’s capsule. [1]
(b) Although kidneys filter a large amount (about 180 L daily) of fluids, still the excretion from the body is only about a litre or two. Why? [1]
(c) What is excretion? Why is it necessary for any living organism? [2]
OR
(c) State two similarities between lungs and kidneys. [2]
Chapter:
In an angiospermic flower, fertilization is said to take place when male gametes carried in the pollen tube fuse with the female gamete, present in the embryo sac. This results in lot of changes in different parts of the flower. State the changes taking place in the following structures of flower post fertilization:
- Zygote
- Ovule
- Ovary
- Sepals
Chapter:
- Sugarcane does not produce seeds so name the process through which it will be able to reproduce.
- List any two advantages of this method.
- Besides sugarcane, give two more examples of plants that reproduce by this method.
- Why regeneration is not possible in all the types of animals?
Chapter:
The volume ratio of hydrogen and oxygen gases liberated during electrolysis of water is ______.
1 : 2
2 : 1
1 : 3
4 : 1
Chapter:
Which of the following set of compounds does not belong to same homologous series?
CH4 and C4H10
C2H6 and C3H8
C3H8 and C5H12
C4H8 and C5H12
Chapter:
Advertisements
Which of the following is an amphoteric oxide?
Na2O
K2O
CO2
Al2O3
Chapter:
Study the following table and select the correct option:
| Salt | Acid used | Base used | Nature of Salt |
| NaCl | HCl | NaOH | Basic |
| Salt | Acid used | Base used | Nature of Salt |
| Na2CO3 | H2CO3 | NaOH | Neutral |
| Salt | Acid used | Base used | Nature of Salt |
| Na2SO4 | H2SO4 | NaOH | Acidic |
| Salt | Acid used | Base used | Nature of Salt |
| CH3COONa | CH3COOH | NaOH | Basic |
Chapter:
Which of the following will not undergo addition reaction?
C4H8
C2H2
C3H8
C2H4
Chapter:
Four solutions, namely glucose, alcohol, hydrochloric acid and sulphuric acid filled in four separate beakers are connected one by one in an electric circuit with a bulb. The solutions in which the bulb will glow when current is passed are ______.
Glucose and alcohol
Alcohol and hydrochloric acid
Glucose and sulphuric acid
Hydrochloric acid and sulphuric acid
Chapter:
Which gas is evolved when magnesium metal reacts with very dilute HNO3?
Oxygen
Nitrogen
Hydrogen
Nitrogen Dioxide
Chapter:
Assertion (A): Reaction of quick lime with water is an exothermic reaction.
Reason (R): A large amount of heat is evolved on the reaction quick lime and water.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
What happens when lead (II) nitrate is heated?
Write balanced chemical equation of the reaction involved in support of your answer.
Chapter:
What happens when iron (II) sulphate is heated?
Write balanced chemical equation of the reaction involved in support of your answer.
Chapter:
What happens when silver chloride is exposed to sunlight?
Write a balanced chemical equation of the reaction involved in support of your answer.
Chapter:
Give the chemical name and formula of Plaster of Paris.
Chapter:
Write the preparation of the following compound with balanced chemical equation:
Plaster of Paris
Chapter:
Give reason:
While diluting an acid, it is recommended that the acid should be added to water.
Chapter:
Give reason:
Baking soda is used as an antacid.
Chapter:
| Alcohol forms a homologous series with general formula CnH2n+1 –OH and –OH group as functional group. Ethanol is commonly called alcohol and is used in alcoholic drinks. It is good solvent, used in medicines, cough syrups, tonics etc. |
(a) Write structural formula and name of 4th member of alcohol homologous series. [1]
(b) What happens when ethanol is heated with alkaline KMnO4? Write chemical equation involved. [1]
(c) Write the chemical equation of reaction of ethanol with ethanoic acid in the presence of concentrated H2SO4. Write the name of this reaction. [2]
OR
(c) What happens when ethanol is heated with excess concentrated sulphuric acid at 443 K? Write chemical equation involved. What is the role of concentrated sulphuric acid in this reaction? [2]
Chapter:
Give reasons for the following:
Ionic compound have generally high melting points and boiling points.
Chapter:
Give reasons for the following:
Solder, an alloy of lead and tin, is used for welding electrical wires.
Chapter:
Give reasons for the following:
Carbon cannot reduce the oxides of Na or Mg.
Chapter:
The reaction of compound ‘X’ with aluminium is used to join railway tracks:
- Identify the compound ‘X’.
- Name the reaction.
- Write the balanced chemical equation of the reaction of compound ‘X’ with aluminium.
Chapter:
Write the balanced chemical equations when a mixture of Cu2O and Cu2S is heated.
Chapter:
Write the balanced chemical equations when ZnS is heated in the presence of oxygen.
Chapter:
Advertisements
Give reasons for the following:
The wires carrying current in homes have a coating of PVC.
Chapter:
Give reasons why copper is used to make hot water tanks and not steel (an alloy of iron).
Chapter: [3] Metals and Non-metals
Show the formation of ionic compound CaO with electron dot structure.
[Atomic number: Ca = 20, O = 8]
Chapter:
Rays from the sun converge at a point 25 cm behind a convex lens. The distance at which an object be placed in front of the lens to get a virtual image, is ______.
20 cm
40 cm
50 cm
More than 50 cm
Chapter:
To restore clear vision in persons whose size of the eye ball has reduced, he/she is suggested to use suitable ______.
Converging lens
Diverging lens
Bifocal lens
Cylindrical lens
Chapter:
Assertion (A): The needle of a compass, kept in an external magnetic field, always aligns itself in the North-South direction of the Earth.
Reason (R): The behaviour of the needle of the compass is similar to the behaviour of a freely suspended bar magnet.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
- Relate the speed of light in the given medium with its optical density.
- Using the information given in the table below, arrange the medium A, B and C in the ascending order of their optical density.
Medium Speed of light (m/s) A 2.25 × 108 B 2 × 108 C 2.08 × 108
Chapter:
How does the change in curvature of the eye lens helps us in the process of seeing the nearby objects clearly?
Chapter:
State the range of the power of accommodation of a normal human eye.
Chapter:
Draw a ray diagram to show the correction of eye defect of an old man who can not see an object placed closer than 1 m from his eye, clearly.
Chapter:
Draw the ray diagram for refraction of light through a glass prism and mark angle of refraction and angle of deviation.
Chapter:
When the path of a light ray refracted through a glass prism is reversed how will the angle of deviation change? Explain.
Chapter:
A ray of light after reflection from a concave mirror becomes parallel to its principal axis. Draw a labelled ray diagram to show this.
Chapter:
What is the difference between virtual images of an object formed by a concave mirror and a convex mirror?
Chapter: [9] Light – Reflection and Refraction
“The value of magnification ‘m’ for a mirror is –2.” Calculate the height of object if the height of image is 20 cm.
Chapter:
Why does an electric bulb become dim when an electric heater in parallel circuit is switched ON?
Chapter:
How to connect three resistors each of resistance 8 Ω, so that the equivalent resistance of the combination is 12 Ω? Draw diagram of the combination and justify your answer.
Chapter:
|
Three students Shweta, Ayesha and Samridhi were performing an experiment to understand the factors on which the resistance of a conductor depends. Each one of them completed electric circuit with the help of a cell, an ammeter, a plug key and wire. Shweta put nichrome wire of length ‘l’ in the circuit and after plugging the key, noted current in the ammeter. Ayesha put nichrome wire of same thickness but twice the length i.e. ‘2l’ in the circuit and after plugging the key, noted current in the ammeter. Samridhi took copper wire of length ‘l’ and same thickness in the circuit and after plugging the key, noted current in the ammeter. |
(a) If the ammeter reading is X ampere with nichrome wire of length ‘l’, then what will be the ammeter reading if the length of nichrome wire is doubled with same area of cross-section?
(b) What happens to the ammeter reading if the area of cross-section of nichrome wire is doubled, keeping the length of wire ‘l’ the same?
(c) Define ‘resistivity’. Write its SI unit. Compare the resistivity of an alloy with its constituents metals.
OR
(c) Give reason:
- Tungsten is used almost exclusively for making the filament of electric lamps.
- Conductors of bread-toasters are made of an alloy rather than a pure metal.
Chapter:
Draw a diagram showing the magnetic field lines around a bar magnet.
Chapter:
Write any two characteristics of magnetic field lines.
Chapter:
The concentric circles representing the magnetic field around a current carrying straight wire become larger and larger as we move away from the wire. How does magnetic field change on these concentric circles?
Chapter:
Draw a diagram to represent the magnetic field lines along the axis of a current carrying solenoid. Mark arrows to show the direction of current in the solenoid and the direction of magnetic field lines.
Chapter: [12] Magnetic Effects of Electric Current
Describe some salient features of the magnetic lines of field concept.
Chapter:
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CBSE previous year question papers Class 10 Science with solutions 2025 - 2026
Previous year Question paper for CBSE Class 10 Science-2026 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Science, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CBSE Class 10.
How CBSE Class 10 Question Paper solutions Help Students ?
• Question paper solutions for Science will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
