Science (English Medium)
Academic Year: 2024-2025
Date & Time: 27th February 2025, 10:30 am
Duration: 3h
Advertisements
General Instructions:
Read the following instructions carefully and follow them:
- This question paper contains 33 questions. All questions are compulsory.
- This question paper is divided into FIVE sections - Section A, B, C, D and E.
- Section A - questions number 1 to 16 are multiple choice type questions. Each question carries 1 mark.
- Section B - questions number 17 to 21 are very short answer type questions. Each question carries 2 marks.
- Section C - questions number 22 to 28 are short answer type questions. Each question carries 3 marks.
- Section D - questions number 29 and 30 are case-based questions. Each question carries 4 marks.
- Section E - questions number 31 to 33 are long answer type questions. Each question carries 5 marks.
- There is no overall choice given in the question paper. However, an internal choice has been provided in few questions in all the sections except Section - A.
- Kindly note that there is a separate question paper for Visually Impaired candidates.
- Use of calculator is NOT allowed.
You may use the following values of physical constants wherever necessary:
c = 3 × 108 m/s
h = 6.63 × 10−34 Js
e = 1.6 × 10−19 C
µ0 = 4π × 10−7 T m A−1
ε0 = 8.854 × 10−12 C2 N−1 m−2
`1/(4πε_0)` = 9 × 109 N m2 C−2
Mass of electron (me) = 9.1 × 10−31 kg
Mass of neutron = 1.673 × 10−27 kg
Avogadro’s number = 6.023 × 1023 per gram mole
Boltzmann’s constant = 1.38 × 10−23 JK−1
The charge required for the reduction of 1 mol of \[\ce{MnO^-_4}\] to MnO2 is ______.
1 F
3 F
5 F
6 F
Chapter:
Which among the following is a false statement?
Rate of zero order reaction is independent of initial concentration of reactant.
Half-life of a zero order reaction is inversely proportional to the rate constant.
Molecularity of a reaction may be zero.
For a first order reaction, t1/2 = 0.693/k.
Chapter:
The number of molecules that react with each other in an elementary reaction is a measure of the ______.
activation energy of the reaction
stoichiometry of the reaction
molecularity of the reaction
order of the reaction
Chapter:
The element having [Ar] 3d104s1 electronic configuration is ______.
Cu
Zn
Cr
Mn
Chapter:
The complex ions [Co(NH3)5 (NO2)]2+ and [Co(NH3)5 ONO)]2+ are called ______.
Ionisation isomers
Linkage isomers
Co-ordination isomers
Geometrical isomers
Chapter:
The diamagnetic species is ______.
[At. No.: Co = 27, Fe = 26, Ni = 28]
[Ni(CN)4]2–
[NiCl4]2–
[Fe(CN)6]3–
[CoF6]3–
Chapter:
Which is the correct IUPAC name for
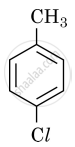
Methylchlorobenzene
Toluene
1-Chloro-4-Methylbenzene
1-Methyl-4-Chlorobenzene
Chapter:
What will be formed after oxidation reaction of secondary alcohol with chromic anhydride (CrO3)?
Aldehyde
Ketone
Carboxylic acid
Ester
Chapter:
The conversion of phenol to salicylic acid can be accomplished by ______.
Reimer-Tiemann reaction
Friedel-Crafts reaction
Kolbe reaction
Coupling reaction
Chapter:
Which of the following is/are examples of denaturation of protein?
Coagulation of egg white
Curdling of milk
Clotting of blood
Both (A) and (B)
Chapter:
Nucleotides are joined together by ______.
Glycosidic linkage
Peptide linkage
Hydrogen bonding
Phosphodiester linkage
Chapter:
Scurvy is caused due to deficiency of ______.
Vitamin B1
Vitamin B2
Ascorbic acid
Glutamic acid
Chapter:
Assertion (A): In a first order reaction, if the concentration of the reactant is doubled, its half-life is also doubled.
Reason (R): The half-life of a reaction does not depend upon the initial concentration of the reactant in a first order reaction.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of Assertion (А).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
Assertion (A): Cu cannot liberate hydrogen from acids.
Reason (R): Because it has positive electrode potential.
Both assertion and reason are true, and reason is the correct explanation of the assertion.
Both assertion and reason are true but reason is not the correct explanation of assertion.
Assertion is not true but reason is true.
Both assertion and reason are false.
Chapter: [4] d-block and f-block Elements
Assertion (A): Aromatic primary amines cannot be prepared by Gabriel Phthalimide synthesis.
Reason (R): Aryl halides do not undergo nucleophilic substitution reaction with the anion formed by phthalimide.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of Assertion (А).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
Assertion (A): Vitamin D cannot be stored in our body.
Reason (R): Vitamin D is fat soluble vitamin and is not excreted from the body in urine.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of Assertion (А).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
The rate constant for a zero order reaction A → P is 0.0030 mol L−1s−1. How long will it take for the initial concentration of A to fall from 0.10 M to 0.075 M?
Chapter:
The decomposition of NH3 on platinum surface is zero order reaction. What are the rates of production of N2 and H2 if k = 2.5 × 10−4 mol−1 L s−1?
Chapter: [3] Chemical Kinetics
Define the following term:
Pseudo first-order reaction
Chapter: [3] Chemical Kinetics
Give reason for the following:
The transition metals generally form coloured compounds.
Chapter: [4] d-block and f-block Elements
Account for the following:
Zn is not considered as a transition element.
Chapter: [4] d-block and f-block Elements
Advertisements
Name the following coordination compound according to IUPAC norm:
[Co(NH3)4(H2O)Cl]Cl2
Chapter:
Name the following coordination compounds according to IUPAC norm:
[CrCl2(en)2]Cl
Chapter:
In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?

Chapter: [6] Haloalkanes and Haloarenes
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
2-Bromo-2-methylbutane, 1-Bromopentane, 2-Bromopentane
Chapter: [6] Haloalkanes and Haloarenes
At 25 °C, the saturated vapour pressure of water is 24 mm Hg. Find the saturated vapour pressure of a 5% aqueous solution of urea at the same temperature. (Molar mass of urea = 60 g mol−1)
Chapter:
The electrical resistance of a column of 0.05 M NaOH solution of area 0.8 cm2 and length 40 cm is 5 × 103 ohm. Calculate its resistivity, conductivity and molar conductivity.
Chapter:
Complete and balance the following chemical equation:
\[\ce{MnO^-4 + C2O^{2-}4 + H+ ->}\]
Chapter:
Complete and balance the following chemical equation:
\[\ce{KMnO4 ->[Heat][513 K]}\]
Chapter:
Complete and balance the following chemical equation:
\[\ce{Cr2O^{2-}7 + H2S + H+ ->}\]
Chapter:
Using valence bond theory, explain the hybridisation and magnetic character of the following:
[Co(NH3)6]3+
Chapter:
Using valence bond theory, predict the hybridization and magnetic character of the complex:
[Ni(CO)4] (Atomic number: Ni = 28)
Chapter:
Define racemic mixture.
Chapter: [6] Haloalkanes and Haloarenes
Why is chlorobenzene resistant to nucleophilic substitution reaction?
Chapter:
Explain the following reaction:
Wolff-Kishner reduction
Chapter: [8] Aldehydes, Ketones and Carboxylic Acids
Explain the following reaction and write the chemical equation involved:
Etard reaction
Chapter:
Explain the following reaction:
Cannizzaro reaction
Chapter: [8] Aldehydes, Ketones and Carboxylic Acids
Write the structure of A, B, and C in the following sequence of reactions:
\[\ce{CH3COOH ->[SOCl2]A ->[H2, Pd-BaSO4][]B ->[H2N-NH2][] C}\]
Chapter:
Write the structure of A, B, and C in the following sequence of reactions:
\[\ce{CH3CN->[{1.} (DIBAL-H)][{2.} H2O] A ->[Dil{.} NaOH][] B ->[Δ][] C}\]
Chapter:
Advertisements
| The spontaneous flow of the solvent through a semipermeable membrane from a pure solvent to а solution or from a dilute solution to a concentrated solution is called osmosis. The phenomenon of osmosis can be demonstrated by taking two eggs of the same size. In an egg, the membrane below the shell and around the egg material is semipermeable. The outer hard shell can be removed by putting the egg in dilute hydrochloric acid. After removing the hard shell, one egg is placed in distilled water and the other in a saturated salt solution. After some time, the egg placed in distilled water swells-up while the egg placed in salt solution shrinks. The external pressure applied to stop the osmosis is termed as osmotic pressure (a colligative property). Reverse osmosis takes place when the applied external pressure becomes larger than the osmotic pressure. |
- Define reverse osmosis. Name one SPM which can be used in the process of reverse osmosis. [2]
-
- What do you expect to happen when red blood corpuscles (RBC’s) are placed in 0.5% NaCl solution? [1]
OR - Which one of the following will have a higher osmotic pressure in 1 M KCl or 1 M urea solution? Justify your answer. [1]
- What do you expect to happen when red blood corpuscles (RBC’s) are placed in 0.5% NaCl solution? [1]
- Why is osmotic pressure a colligative property? [1]
Chapter:
| Amines have a lone pair of electrons on nitrogen atom due to which they behave as Lewis base. Greater the value of Kb or smaller the value of pKb, stronger is the base. Amines are more basic than alcohols, ethers, esters, etc. The basic character of aliphatic amines should increase with the increase of alkyl substitution. But it does not occur in a regular manner as a secondary aliphatic amine is unexpectedly more basic than a tertiary amine in aqueous solutions. Aromatic amines are weaker bases than ammonia and aliphatic amines. Electron releasing groups such as −CH2, −OCH3, −NH2, etc., increase the basicity while electron-withdrawing substituents such as −NO2, −CN, halogens, etc., decrease the basicity of amines. The effect of these substitute is more at p− than at m− position. |
- Arrange the following in the increasing order of their basic character. Give reason: [2]
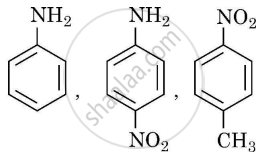
- Why pKb of aniline is more than that of methylamine? [1]
-
- Arrange the following in the increasing order of their basic character in an aqueous solution: [1]
(CH3)3N, (CH3)2NH, NH3, CH3NH2
OR - Why is ammonolysis of alkyl halides not a good method to prepare pure amines? [1]
- Arrange the following in the increasing order of their basic character in an aqueous solution: [1]
Chapter:
Give the IUPAC name of the following compound.
CH3 − CH = CH − CHO
Chapter: [8] Aldehydes, Ketones and Carboxylic Acids
Propanal and Propanone
Chapter: [8] Aldehydes, Ketones and Carboxylic Acids
How do you convert toluene to benzoic acid?
Chapter: [8] Aldehydes, Ketones and Carboxylic Acids
How do you convert the following:
Ethanol to Propan-2-ol
Chapter: [7] Alcohols, Phenols and Ethers
How will you convert the following:
Propanal to 2-hydroxy propanoic acid
Chapter:
Complete the synthesis by giving missing starting material, reagent or product.
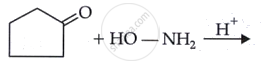
Chapter:
Complete the synthesis by giving missing starting material, reagent or product.
![]()
Chapter: [8] Aldehydes, Ketones and Carboxylic Acids
Complete the synthesis by giving missing starting material, reagent or product.
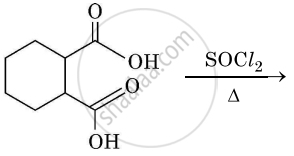
Chapter:
Complete the synthesis by giving missing starting material, reagent or product.
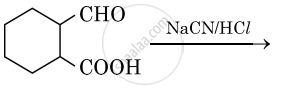
Chapter:
Complete synthesis by giving missing starting material, reagent or product:
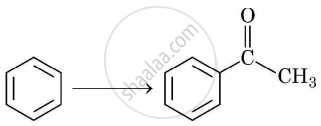
Chapter:
Calculate the standard Gibbs energy (ΔrG°) of the following reaction at 25°C:
\[\ce{Au_{(s)} + Ca^2 + (1M) ->Au^3 + (1M) + Ca_{(s)}}\]
`E_(Au^(3+)"/"Au)^° = +1.5 V, E_(Ca^(2+)"/"Ca)^° = -2.87 V`
Predict whether the reaction will be spontaneous or not at 25 °C. [1 F = 96500 C mol−1]
Chapter:
Tarnished silver contains AgS. Can this tarnish be removed by placing tarnished silverware in an aluminium pan containing an inert electrolytic solution such as NaCl? The standard electrode potential for half reaction:
\[\ce{Ag2S_{(s)} + 2e- -> 2Ag_{(s)} + S^{2-}}\] is −0.71 V and for \[\ce{Al^{3+} + 3e- -> 2Al_{(s)}}\] is −1.66 V.
Chapter:
Define the following term:
Fuel cell
Chapter: [2] Electrochemistry
Calculate the emf of the cell reaction at 25°C:
\[\ce{Zn_{(s)}_{(0.1 M)} {|} Zn^{2+}_{ (aq)}_{(0.1 M)} {||} Cd^{2+}_{ (aq)}_{(0.01 M)} {|} Cd_{(s)}}\]
Given: `E_(Cd^(2+)//Cd)^° = −0.40 V`
`E_(Zn^(2+)//Zn)^°= −0.76 V`
[log 10 = 1]
Chapter:
An organic compound ‘A’, molecular formula C2H6O oxidises with CrO3 to form a compound ‘B’. Compound ‘B’ on warming with iodine and aqueous solution of NaOH gives a yellow precipitate of compound ‘C’. When compound ‘A’ is heated with conc. H2SO4 at 413 K gives a compound ‘D’, which, on reaction with excess HI gives compound ‘E’. Identify compounds ‘A’, ‘B’, ‘C’, ‘D’ and ‘E’, and write the chemical equations involved.
Chapter:
Write the chemical equation of the following reaction:
Phenol is treated with conc. HNO3.
Chapter:
Write the chemical equation of the following reaction:
Propene is treated with B2H followed by oxidation by H2О2/ОН−.
Chapter:
Write the chemical equation of the following reaction:
Sodium t-butoxide is treated with CH3Cl.
Chapter:
Give a simple chemical test to distinguish between butan–1–ol and butan–2–ol.
Chapter:
Arrange the following in increasing order of acid strength:
phenol, ethanol, water
Chapter:
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CBSE previous year question papers Class 12 Chemistry with solutions 2024 - 2025
Previous year Question paper for CBSE Class 12 -2025 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Chemistry, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CBSE Class 12.
How CBSE Class 12 Question Paper solutions Help Students ?
• Question paper solutions for Chemistry will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
