Advertisements
Advertisements
प्रश्न
Write the structure of the product of the following reaction:
\[\ce{H3C - C ≡ C - H ->[Hg^{2+}, H2SO4]}\]
Advertisements
उत्तर
\[\begin{array}{cc}
\phantom{..............................}\ce{OH}\phantom{..................}\ce{O}\\
\phantom{..............................}|\phantom{.....................}||\phantom{}\\
\ce{H3C - \underset{Propyne}{C ≡ C} - H ->[Hg^{2+}, H2SO4] [CH3 - C = CH2] -> CH3 \underset{Propanone}{- C -} CH3}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
What is the action of Zn – Hg / conc. HCl on propanone?
How will you convert calcium acetate to acetaldehyde?
What is the action of following reagents on ethanoic acid?
SOCl2/heat
What is the action of following reagents on ethanoic acid?
P2O5/heat
Which of the following is a Stephen reaction?
The reaction in which methyl group on benzene ring is converted to aldehydic group is called ______.
A reaction in which chromyl chloride in presence of CS2 converts methyl group of toluene to aldehydic group on acid hydrolysis is called _________.
Which of the following compounds does NOT undergo Friedel-Crafts reaction?
Identify 'A' in the following reaction.
\[\ce{A ->[H2][Pd-BaSO4] C6H5CHO + HCl}\]
Which of the following reaction is shown by alkynes?
The reaction

How ketones are prepared from nitriles?
What is the action of hydrogen cyanide on the following?
Benzaldehyde
Write Stephen reaction for the preparation of aldehyde.
How are the following compound obtained by hydration of alkenes?
Ethyl alcohol
How are the following compound obtained by hydration of alkenes?
Isopropyl alcohol
Write the name of the product formed when ethanoyl chloride is treated with dimethyl cadmium.
Which of the following on heating with aqueous KOH produces acetaldehyde.
Identify the product when alkyl alkanoate is hydrolysed with dil. HCl obtained when.
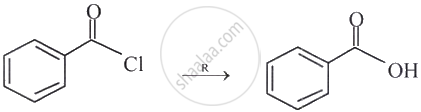
Identify the reagent ‘R’ necessary to bring the following conversion.