Advertisements
Advertisements
प्रश्न
Write the chemical formula of the following compound in a step-by-step manner –
Potassium permanganate
Advertisements
उत्तर
Write the formula of
| Potassium permanganate | Symbol | valency |
| Potassium | \[\ce{K}\] | \[\ce{1^+}\] |
| permanganate | \[\ce{MnO4}\] | \[\ce{1^-}\] |
Step I – Write each symbol with its valency.
Positive ion is written first
\[\ce{K}^{1+}\] \[\ce{[MnO4]}^{1-}\]
Step II – Interchange the valencies and ignoring the (+) and (−) signs.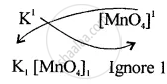
The Formula is \[\ce{KMnO4}\]
APPEARS IN
संबंधित प्रश्न
Write the molecular formula of the following compounds of Sulphur trioxide.
Find the number and names of elements present in them and calculate their molecular masses.
Write the molecular formulae for the following compounds and name the elements present of Nitric acid.
The valency of aluminium is 3. Write the valency of other radicals present in the following compounds of Aluminium oxide.
The valency of aluminium is 3. Write the valency of other radicals present in the following compounds of Aluminium sulphate.
What is variable valency ? Give two examples of elements showing variable valency.
Copper having electronic configuration 2,8,18,1 exhibits variable valency. Give a reason for the same and name the compound CuCI and CuCl2.
With reference to water and ammonia as compounds respectively, state the valency of oxygen and nitrogen.
Write the chemical formula of the following compound in a step-by-step manner –
Zinc hydroxide
Write the chemical formula of the following compound in a step-by-step manner –
Copper [I] sulphide
Match the statements – 1 to 10 below with their correct answers from – A to J.
| Column I | Column II |
| 1. Elements having valency of two. | A: Br1− |
| 2. An anion | B: Divalent |
| 3. A gaseous non-metal | C: Reactants |
| 4. A cation. | D: Ammonium |
| 5. The term used for the substances which take part in the chemical reaction | E: Nitric oxide |
| 6. The meaning of the symbol ‘Δ’ over the arrow in a chemical equation. | F: Nitrogen |
| 7. The chemical name for nitrogen monoxide. | G: Zero |
| 8. A radical containing nitrogen and hydrogen only | H: Nitrous oxide |
| 9. The chemical name for dinitrogen oxide | I: Heat required |
| 10. The valency of noble gases | J: K1+ |
