Advertisements
Advertisements
प्रश्न
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagents :
CH3MgBr and then H3O+
Advertisements
उत्तर
Acetone reduces to form tertiary alcohol in presence of grignard reagent.

संबंधित प्रश्न
Name the reagents used in the following reactions:

Name the reagents used in the following reactions:

How is the following conversion carried out?
\[\ce{Benzyl chloride -> Benzyl alcohol}\]
How is the following conversion carried out?
\[\ce{Methyl magnesium bromide -> 2-Methylpropan-2-ol}\]
Name the reagent used in the following reaction:
Dehydration of propan-2-ol to propene.

Show how you would synthesise the following alcohol from an appropriate alkene?
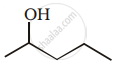
Which of the following reacts with NaOH to give alcohol?
When glycol is heated with dicorboxylic acid the product are
The major product of the following reaction is:
\[\begin{array}{cc}
\ce{Cl}\phantom{.........................}\\
|\phantom{..........................}\\
\ce{CH3 - CH - CH3 ->[(i) Alc. KOH][(ii) HBr/peroxide (iii) aq. KOH]}
\end{array}\]
