Advertisements
Advertisements
प्रश्न
Write a short note on Wurtz-Fittig reaction.
रासायनिक समीकरण/संरचनाएँ
टिप्पणी लिखिए
Advertisements
उत्तर
- The Wurtz-Fittig reaction is a hybrid coupling reaction between an aryl halide and an alkyl halide.
- It involves the use of sodium metal as a reducing agent.
- Sodium metal facilitates the removal of halogen atoms from both aryl and alkyl halides.
- This results in the formation of a new carbon-carbon bond between an aromatic ring and an alkyl group.
- The reaction produces alkylated aromatic compounds.
- It is carried out under anhydrous conditions typically with heating.
- The mechanism involves the formation of intermediate organosodium compounds or radicals that couple to give the product.
- The Wurtz-Fittig reaction is useful for synthesizing alkyl-substituted aromatic compounds, combining features of Wurtz reaction (alkyl-alkyl coupling) and Fittig reaction (aryl-aryl coupling).
- When a haloarene is heated with an ethereal solution of an alkyl halide in the presence of sodium, the alkyl group replaces the halogen atom of the haloarene, resulting in a higher arene. The Wurtz-Fittig process is used to prepare higher homologues of benzene.
For example:
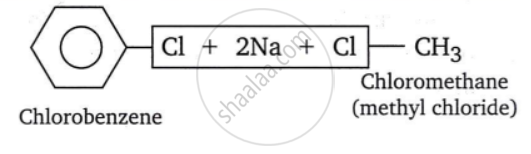

shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
