Advertisements
Advertisements
प्रश्न
Which of the following graphs is not correct for ideal gas?
विकल्प
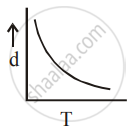
[d = density, P = pressure, T = Temperature]
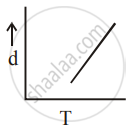
[d = density, P = pressure, T = Temperature]
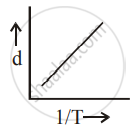
[d = density, P = pressure, T = Temperature]
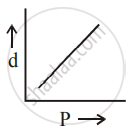
[d = density, P = pressure, T = Temperature]
MCQ
Advertisements
उत्तर

[d = density, P = pressure, T = Temperature]
Explanation:
From the ideal gas equation, we get,
PM = dRT
⇒ d = `["PM"/"R"]1/"T"`
So graph d vs T is not a straight line.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
