Advertisements
Advertisements
प्रश्न
What do you observe when Excess ammonia is mixed with chlorine.
एक पंक्ति में उत्तर
Advertisements
उत्तर
When excess ammonia is mixed with chlorine, ammonium chloride and
nitrogen is formed.
\[\ce{8NH3(excess) + 3CI2 -> N2 + 6NH4CI}\]
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
APPEARS IN
संबंधित प्रश्न
Is ammonia more dense or less dense than air?
How ammonia occurs in nature?
Give the formula of (i) Liquid ammonia (ii) Liquor ammonia
What is liquor ammonia fortis?
When an ammonium salt is warmed with sodium hydroxide solution, ammonia gas is evolved. State three ways by which you can identify this gas.
Ammonium salt decompose on heating. What other property do ammonium salts have in common?
Name the gas evolved when the following mixtures are heated.
Calcium hydroxide and ammonium chloride
The diagram shows an experimental set up for the laboratory preparation of a pungent smelling gas. The gas is alkaline in nature.
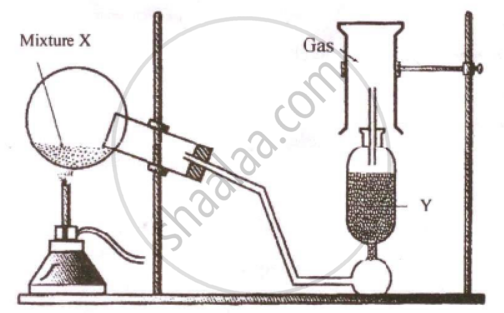
- Nature the gas collected in the jar.
- Write the balance equation for the above preparation.
- How is the gas being collected?
- Name the drying agent used.
- How will you find that the jar is full of gas?
What do you observe when Ammonia is passed over heated lead oxide.
What do you observe when Ammonia comes in contact with the eyes of a person.
