Advertisements
Advertisements
प्रश्न
The preparation of alkyl fluoride from alkyl chloride, in presence of metallic fluorides is known as ______________.
विकल्प
Williamson’s reaction
Finkelstein reaction
Swarts reaction
Wurtz reaction
Advertisements
उत्तर
Swarts reaction
Preparation of alkyl fluorides from alkyl chlorides or bromides in presence of metallic fluorides like AgF, Hg2F2 reaction is known as Swarts reaction.
R - X + AgF → R - F + AgX
APPEARS IN
संबंधित प्रश्न
How is propene converted into 1- bromopropane and 2 - bromopropane?
What is the action of bromine in alkaline medium on
i. CH3CH2NO2
ii. 
Draw the structures of major monohalo products in each of the following reactions :

Draw the structure of major monohalo product in each of the following reactions :
Write a short note on Sandmeyer’s reaction.
The synthesis of alkyl fluorides is best accomplished by ____________.
Gem-dibromide is ____________.
The catalyst used in the preparation of an alkyl chloride by the action of dry HCl on alcohol is ____________.
The order of reactivity of alcohols with halogen acids is ______.
(A) \[\ce{CH3CH2 - CH2 - OH}\]
(B) \[\begin{array}{cc}
\phantom{}\ce{CH3CH2 - CH - OH}\\
\phantom{...}\phantom{}|\\
\phantom{......}\ce{CH3}
\end{array}\]
(C) \[\begin{array}{cc}
\phantom{........}\ce{CH3}\\
\phantom{.....}\phantom{}|\\
\phantom{}\ce{CH3CH2 - C - OH}\\
\phantom{.....}\phantom{}|\\
\phantom{........}\ce{CH3}
\end{array}\]
Which of the following alcohols will yield the corresponding alkyl chloride on reaction with concentrated HCl at room temperature?
Alkyl halides are prepared from alcohol by treating with ______.
(i) HCl + ZnCl2
(ii) Red P + Br
(iii) H2SO4 + KI
(iv) All the above
Alkyl fluorides are synthesised by heating an alkyl chloride/bromide in presence of ______ or ______.
(i) CaF2
(ii) CoF2
(ii) Hg2F2
(iv) NaF
Aryl chlorides and bromides can be easily prepared by electrophilic substitution of arenes with chlorine and bromine respectively in the presence of Lewis acid catalysts. But why does preparation of aryl iodides requires presence of an oxidising agent?
Discuss the role of Lewis acids in the preparation of aryl bromides and chlorides in the dark.
tert-Butylbromide reacts with aq. \[\ce{NaOH}\] by SN1 mechanism while n-butylbromide reacts by SN2 mechanism. Why?
Assertion: Phosphorus chlorides (tri and penta) are preferred over thionyl chloride for the preparation of alkyl chlorides from alcohols.
Reason: Phosphorus chlorides give pure alkyl halides.
Which of the following is the most stable free radical?
The alky halide is converted into an alcohol by
Which is gem-dihalide?
Benzyl chloride (16H5CH2Cl) can be prepared from toluene by chlorination with
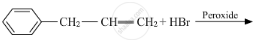
The major product of the following reaction is:
\[\ce{C2H5Cl + AgF -> C2H5F + AgCl}\] The above reaction is called ______.
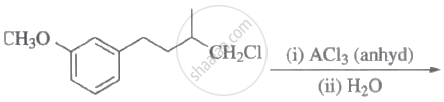
The major product of the following reaction is:
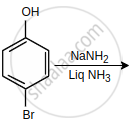
In the given reactions sequence, the major product 'C' is:
\[\ce{C8H10 ->[HNO3][H2SO4] A ->[Br2][\Delta] B ->[alcoholic][KOH] C}\]
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
