Advertisements
Advertisements
प्रश्न
How is propene converted into 1- bromopropane and 2 - bromopropane?
Advertisements
उत्तर
The addition of hydrogen halide to an unsymmetrical alkene gives two products.
Propene on reaction with hydrogen bromide forms 80% 2-bromopropane (isopropyl bromide) and 20% 1-bromopropane (n-propyl bromide).

APPEARS IN
संबंधित प्रश्न
Draw the structures of major monohalo products in each of the following reactions :

In which of the following compound yield is high?
Finkelstein reaction is ______.
Conant Finkelstein reaction for the preparation of alkyl iodide is based upon the fact that:
The boiling points of alcohols are higher than those of hydrocarbons of comparable masses due to ______.
The order of reactivity of alcohols with halogen acids is ______.
(A) \[\ce{CH3CH2 - CH2 - OH}\]
(B) \[\begin{array}{cc}
\phantom{}\ce{CH3CH2 - CH - OH}\\
\phantom{...}\phantom{}|\\
\phantom{......}\ce{CH3}
\end{array}\]
(C) \[\begin{array}{cc}
\phantom{........}\ce{CH3}\\
\phantom{.....}\phantom{}|\\
\phantom{}\ce{CH3CH2 - C - OH}\\
\phantom{.....}\phantom{}|\\
\phantom{........}\ce{CH3}
\end{array}\]
Which of the following alcohols will yield the corresponding alkyl chloride on reaction with concentrated HCl at room temperature?
Which of the following are secondary bromides?
(i) \[\ce{(CH3)2 CHBr}\]
(ii) \[\ce{(CH3)3C CH2Br}\]
(iii) \[\ce{CH3CH(Br)CH2CH3}\]
(iv) \[\ce{(CH3)2 CBrCH2CH3}\]
Alkyl halides are prepared from alcohol by treating with ______.
(i) HCl + ZnCl2
(ii) Red P + Br
(iii) H2SO4 + KI
(iv) All the above
Write down the structure and IUPAC name for neo-pentylbromide.
Match the structures given in Column I with the names in Column II.
| Column I | Column II | |
| (i) |  |
(a) 4-Bromopent-2-ene |
| (ii) | 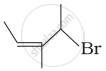 |
(b) 4-Bromo-3-methylpent-2-ene |
| (iii) | 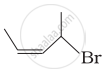 |
(c) 1-Bromo-2-methylbut-2-ene |
| (iv) | 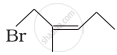 |
(d) 1-Bromo-2-methylpent-2-ene |
Which of the following species is an odd electron intermediate?
The most stable free radical among the following is
The alky halide is converted into an alcohol by
Which is gem-dihalide?
Benzoyl chloride is is prepared from benzoic acid by
SN1 and SN2 product are same with
PClS react with propanone, to give:
Predict the reagent for carrying out the following transformations:
Ethanoic acid to 2-chloroethanoic acid
The major product of the following reaction is:
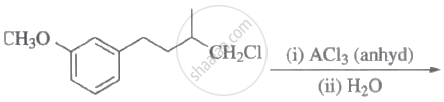
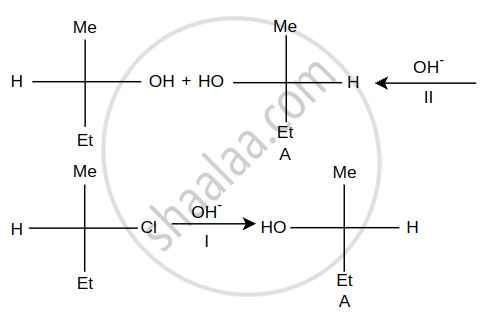
Steps I and II are ______.
\[\ce{C2H5Cl + AgF -> C2H5F + AgCl}\] The above reaction is called ______.
The major product of the following reaction is:
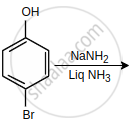
The decreasing order of reactivity of the following organic molecules towards AgNO3 solution is ______.
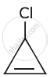

- \[\begin{array}{cc}\ce{CH3CHCH3}\\
|\phantom{..}\\\ce{Cl}\phantom{.}\end{array}\] - \[\begin{array}{cc}\ce{CH3CHCH2NO2}\\
|\phantom{......}\\\ce{Cl}\phantom{.....}\end{array}\]
