Advertisements
Advertisements
प्रश्न
Study the experimental setup in the figure and then answer the questions that follow.
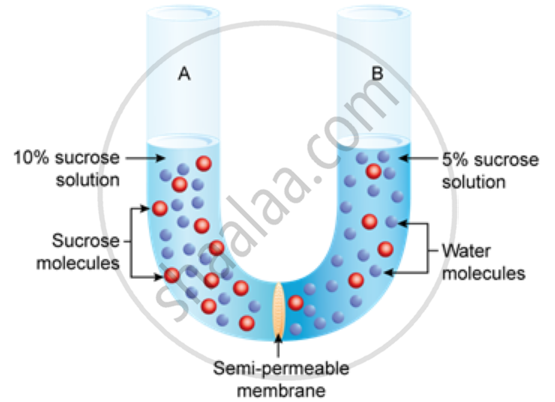
- What phenomenon is being studied by this setup?
- Explain the phenomenon mentioned in (a) above.
- What is meant by 'semipermeable membrane'?
- What will you observe in the set-up after about half an hour? Give a reason for your answer.
संक्षेप में उत्तर
Advertisements
उत्तर
- Osmosis
- Water molecules are transported from their region of higher concentration (dilute solution or with a lower solute concentration) to their region of lower concentration (concentrated solution or with a higher solute concentration) via a semi-permeable membrane. This process is known as osmosis.
- Because it lets just the solvent particles pass through it rather than the solutes, the membrane is known as semipermeable.
- Osmosis will keep water molecules passing from a 5% sucrose solution to 10% sucrose solution over the semipermeable membrane. This will keep on until the water molecule concentration at both ends of the arrangement equal each other.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
