Advertisements
Advertisements
प्रश्न
State three factors on which the rate of emission of electrons from a metal surface depends
Advertisements
उत्तर १
A rate of emission of electrons from a metal surface depends on
(1) Nature of the metal surface: Lower the work function of the metal, greater is the rate of emission of electrons from its surface.
(2) The temperature of the surface: Higher the temperature of the surface, greater is the rate of emission of electrons from the surface.
(3) Surface area of the metal: Larger the surface area of the metal emitting electrons, greater is the rate of emission of electrons.
उत्तर २
(i) The material of the surface,
(ii) The temperature of the surface,
(iii) The surface area of the surface.
APPEARS IN
संबंधित प्रश्न
Arrange α, β, and γ rays in ascending order with respect to their
1) Penetrating power.
2) Ionising power
3) Biological effect
State the energy conversion taking place in a cathode ray tube
What happens if the negative potential is changed on a grid?
A nucleus `""_11^24Na` emits a beta particle to change into Magnesium (Mg)
(i) Write the symbolic equation for the process.
(ii) What are numbers 24 and 11 called?
(iii) What is the general name of `""_12^24Mg `with respect to `""_11^24Na` ?
What do you mean by Atomic mass
Give the relative ionizing power of α, β and γ radiations.
In the following fig. shows a radioactive source S in a thick lead container. The radiations pass through an electric field between the plates P and Q. Complete the diagram to show the paths of α , β and γ radiations.
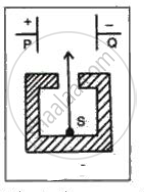
Why is the source S placed in a thick lead container?
State three safety precautions that you would take while handling the radioactive substances.
The half-life of a radioactive nuclide is 100 hours. The fraction of original activity that will remain after 150 hours would be ______.
In the following atoms, which one is a radioisotope? Give one use of this isotope.
O16, C14, N14, He4
