Advertisements
Advertisements
प्रश्न
State one relevant observation for given reactions:
Action of dilute Hydrochloric acid on iron (II) sulfide.
Advertisements
उत्तर
Dilute hydrochloric acid decomposes iron(II) sulfide to produce iron(II) chloride and hydrogen sulfide having rotten egg smell.
FeS + 2HCl → FeCl2 + H2S
APPEARS IN
संबंधित प्रश्न
Potassium sulfite on reacting with hydrochloric acid releases _______ gas. (Cl2, SO2, H2S)
State your observation when dilute hydrochloric acid is added to a lead nitrate solution and the mixture is heated.
Dry hydrogen chloride gas can be collected by ________ displacement of air. (downward / upward)
Give reason for the following:
An aqueous solution of chlorine in acidic in nature
Hint : Cl2 + H2O ⟶ HCl +HClO
State the use of aqua-regia.
What is the function of HCI in preparation of aqua-regia?
Fill in the blank:
On addition of silver nitrate to hydrochloric acid ___________ precipitate is formed which is soluble in ____________
State your observation when:
Decomposition of bicarbonates by dil. H2SO4
2NaHCO3 + H2SO4 → Na2SO4 + 2H2O + 2CO2
2KHCO3 + H2SO4 → K2SO4 + 2H2O + 2CO2
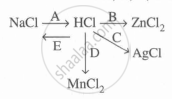
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
