Advertisements
Advertisements
प्रश्न
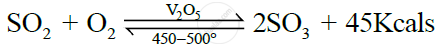
State the conditions required for the given reaction to take place:
Any two conditions for the conversion of sulphur dioxide to sulphur trioxide
Advertisements
उत्तर
Two conditions for the conversion of sulphur dioxide to sulphur trioxide is as follows: Temperature: 450–500° C Catalyst: V2O5
APPEARS IN
संबंधित प्रश्न
State one relevant observation for given reactions:
Action of concentrated Sulphuric acid on hydrated copper sulfate
Give balanced chemical equations for the action of sulphuric acid on the following:
Sulphur
Which property of sulphuric acid is shown by the reaction of the concentrated sulphuric acid with Ethanol?
Name the products formed when hot and concentrated sulphuric acid reacts with NaOH.
Define the following term : Dehydrating agent
Give reason for the following:
Ammonia gas cannot be dried by passing through concentrated sulphuric acid.
Write balanced chemical equation to show :
The behavior of H2SO4 as an acid when it reacts with magnesium.
Name the acid formed when sulphur dioxide dissolves in water.
A, B and C are the chemical properties of sulphuric acid:
A. Oxidizing agent
B. Dehydrating agent
C. Non-volatile acid
Match the following equations 1 to 3 to the above chemical properties of sulphuric acid.
1. \[\ce{KNO3 + H2SO4 -> KHSO4 + HNO3}\]
2. \[\ce{C12H22O11 -> 12C + 11H2O}\]
3. \[\ce{S + 2H2SO4 -> 2H2O + 3SO2}\]
Convert the following reaction into a balanced chemical equation:
Sodium hydroxide to sodium sulphate using sulphuric acid.
