Advertisements
Advertisements
प्रश्न
Solve the following problem.
An electric kettle takes 20 minutes to heat a certain quantity of water from 0 °C to boiling point. It requires 90 minutes to turn all the water at 100 °C into steam. Find the latent heat of vaporization. (Specific heat of water = 1 cal/g °C)
Advertisements
उत्तर
Let heat supplied by the kettle in 20 minutes be Q1 and that in 90 min. be Q2.
Using heat Q1, the temperature of the water is raised from 0 °C to 100 °C.
If the mass of water in the kettle is ‘m’ then,
`"Q"_1 = "ms"_"water" Delta"T" = "m" xx 1 xx (100 - 0)` = 100 m .....(1) .....`(because "S"_"water" = 1 "cal"//"g" °"C")`
Similarly using heat Q2 water is converted from liquid to gas,
∴ Q2 = m Lvap .…(ii)
Given that heat Q1, Q2 are supplied to water in 20 min. (t1) and 90 min (t2) respectively.
Kettle being same its conduction rate (Pcond) is same.
Using, `"P"_"cond" = "Q"_1/"t"_1 = "Q"_2/"t"_2` ...(iii)
From (i), (ii) and (iii),
`"100 m"/20 = "mL"_"vap"/90`
∴ `"L"_"vap" = 5 xx 90 = 450` cal/g
Latent heat of vaporisation for water is 450 cal/ g.
APPEARS IN
संबंधित प्रश्न
Answer the following question.
Explain the term 'steady state'.
Answer the following question.
Define coefficient of thermal conductivity.
Answer the following question.
Derive its expression for the coefficient of thermal conductivity.
Answer the following question.
Explain the term thermal resistance. State its SI unit and dimensions.
Answer the following question.
Which materials can be used as thermal insulators and why?
Solve the following problem.
Which will require more energy, heating a 2.0 kg block of lead by 30 K or heating a 4.0 kg block of copper by 5 K?
(slead = 128 J kg–1 K–1, `"s"_"copper"` = 387 J kg–1 K–1)
Solve the following problem.
Find the temperature difference between two sides of a steel plate 4 cm thick, when the heat is transmitted through the plate at the rate of 400 k cal per minute per square meter at a steady state. Thermal conductivity of steel is 0.026 kcal/m s K.
If 1 g of a steam which is at 100°C starts melting then how much ice will remain at 0° C? (Latent heat of ice = 80 cal/g and latent heat of steam= 540 cal/g)
In order that the heat flows from one part of a solid to another part, what is required?
Heat energy is incident on the surface at the rate of 1000 J/min. If the coefficient of absorption is 0.8 and the coefficient of reflection is 0.1 then heat energy transmitted by the surface in 5 minutes is ______.
A slab consist of two parallel layers of copper and brass of the same thickness and having thermal conductivities in the ratio 1 : 4. If the free face of brass is at 100°C and that of copper at 0°C, the temperature of interface is ______.
Two rectangular blocks A and B of different metals have same length and same area of cross-section. They are kept in such a way that their cross-sectional area touch each other. The temperature at one end of A is 100°C and that of B at the other end is 0°C. If the ratio of their thermal conductivity is 1 : 3, then under steady state, the temperature of the junction in contact will be ______.
A thin square steel plate with each side equal to 10 cm is heated by a blacksmith. The rate of radiated energy by the heated plate is 1134 watts. The temperature of the hot steel plate is ______.
(Stefan's constant σ = 5.67 × 10-8 watt m-2K-4 emissivity of the plate = 1)
An electric heater is used in a room with a total wall area of 137 m2 to maintain a temperature of +20°C inside it when the outside temperature is -10°C. The walls have three different layers of materials. The innermost layer is of wood of thickness 2.5 cm, the middle layer is of cement of thickness 1.0 cm and the outermost layer is brick of 25.0 cm. The power of the electric heater will be ______ W. Assume that there is no heat loss through the floor and the ceiling. The thermal conductivities of wood, cement, and brick are 0.125, 1.5, and 1.0 watt/m/°C respectively.
In the amount of water of mass 20 g at 0°C is mixed with 40 g of water at 10°C, the final temperature of the mixture is ______.
Two rods of the same material, same length and same radius transfer a given amount of heat in t second when they are joined end-to-end. When the rods are joined one above the other, then they will transfer the same heat in the same conditions in time ______.
Through which mode does heat transfer take place in solids?
In which mode of heat transfer do the molecules physically move from one place to another?
At what speed do electromagnetic waves travel in space during radiation?
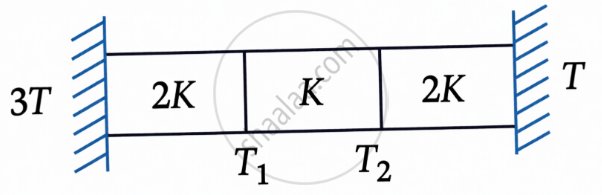
Three identical heat conducting rods are connected in series as shown in the figure. The rods on the sides have thermal conductivity 2K while that in the middle has thermal conductivity K. The left end of the combination is maintained at temperature 3T and the right end the at T. The rods are thermally insulated from outside. In steady state, temperature at the left junction is T1 and that the right junction is T2. The ratio T1/2 T is: