Advertisements
Advertisements
प्रश्न
In the HCl molecule, the separation between the nuclei of the two atoms is about 1.27 Å (1 Å = 10–10 m). Find the approximate location of the CM of the molecule, given that a chlorine atom is about 35.5 times as massive as a hydrogen atom and nearly all the mass of an atom is concentrated in its nucleus.
Advertisements
उत्तर
Let us choose the nucleus of the hydrogen atom as the origin for measuring distance. Mass of hydrogen atom,m1= 1 unit (say) Since chlorine atom is 35.5 times as massive as hydrogen atom,
∴ mass of the cholorine atom, m2 = 35.5 units
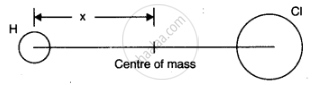
Now, `x_1 = 0 " and " x_2 = 1.27 Å = 1.27 xx 10^(-10) m`
distance of centre of mass of HCL molecule from its origin is given by
`X = (m_1x_1+m_2x_2)/(m_1+m_2) = (1xx0+ 35.5 xx 1.27 xx 10^(-10))/(1+35.5) m`
`= (35.5 xx 1.27)/36.5 xx 10^(-10) m`
`= 1.235 xx 10^(-10) m`
`= 1.235 Å`
