Advertisements
Advertisements
प्रश्न
In following Figure shows a mixed source R of alpha and beta particles in a thick lead walled container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper inwards as shown by ×.
- Show in the diagram how the particles get affected.
- Name the law used in part (a).
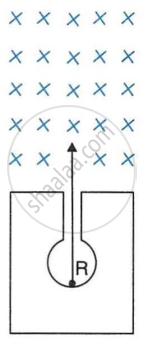
[Hint: Alpha particles will deflect to the left while beta particles to the right]
Advertisements
उत्तर
(a)
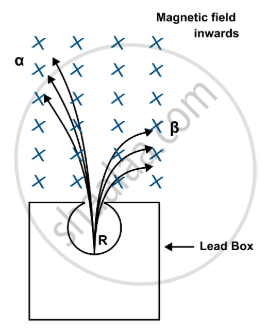
(b)
Alpha particles (α): positively charged → will be deflected to the left.
Beta particles (β): negatively charged → will be deflected to the right.
The direction of deflection in a magnetic field is given by Fleming’s left hand rule.
-
Thumb → direction of force (deflection)
-
Forefinger → direction of magnetic field (into the page)
-
Middle finger → direction of current or moving charge (upward).
APPEARS IN
संबंधित प्रश्न
The diagram in figure shows a radioactive source S placed in a thick lead walled container. The radiations given out are allowed to pass through a magnetic field. The magnetic field (shown as ×) acts perpendicular to the plane of paper inwards. Arrows shows the paths of the radiation A, B and C.
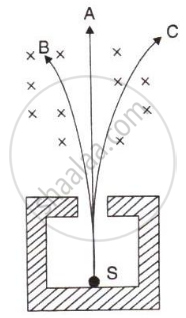
- Name the radiations labelled A, B and C.
- Explain clearly how you used the diagram to arrive at the answer in part (a).
In following Figure shows a radioactive source S in a thick lead walled container having a narrow opening. The radiations pass through an electric field between the plates A and B.
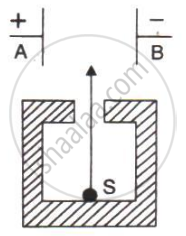
- Complete the diagram to show the paths of α, β and γ radiations.
- Why is the source S kept in a thick lead walled container with a narrow opening?
- Name the radiation which is unaffected by the electrostatic field.
- Which radiation is defleced the most. Given reason.
- Which among the three radiations causes the least biological damage?
Is it possible to deflect γ - radiations in a way similar to α and β -particles, using the electric or magnetic field? Give reasons.
State following four properties each of α, β and γ radiations:
- Nature,
- Charge,
- Mass and
- Effect of electric field.
State the speed of each of α, β and γ radiations.
What is the composition of α, β and γ radiations?
A radioactive substance emits radiations ______.
Compare the penetrating powers of α, β and γ-radiations.
What are α-and β-radiations?
Which nuclear radiation will travel undeviated in an electric field?
