Advertisements
Advertisements
प्रश्न
How is phenol prepared from isopropyl benzene?
Write chemical reactions for the preparation of phenol from isopropyl benzene.
Advertisements
उत्तर
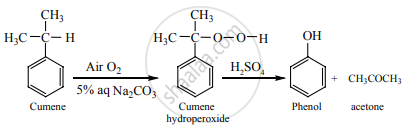
On passing air to a mixture of cumene (isopropyl benzene) and 5% aqueous sodium carbonate solution, cumene hydro peroxide is formed by oxidation. It is treated with dilute acid to get phenol and acetone.
Notes
Students should refer to the answer according to their questions.
संबंधित प्रश्न
Identify the product(s) is/are formed when 1-methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
Write the structure of the aldehyde, carboxylic acid and ester that yield 4-methylpent-2-en-1-ol.
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI
What will be the product (X and A)for the following reaction
acetylchloride \[\ce{{acetylchloride}->[i)CH3MgBr][ii)H3O+]X ->[acidK2Cr2O7] A}\]
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in each of the following method.
Acid catalysed hydration
What will be the product (X and A) for the following reaction
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O^+ ]X ->[acid K2Cr2O7]A}\]
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
The correct IUPAC name of the compound,
\[\begin{array}{cc}
\ce{CH3}\phantom{......}\\
|\phantom{........}\\
\ce{H3C - CH - CH - CH - CH2 - OH}\\
|\phantom{............}|\phantom{........}\\
\ce{Cl}\phantom{...........}\ce{CH3}\phantom{......}
\end{array}\]
