Advertisements
Advertisements
प्रश्न
Give three differences between metals and non-metals.
Advertisements
उत्तर
Metals:
1. Metals are ductile i.e. can be drawn into wires.
2. Metals are malleable i.e. can be beaten to form sheets.
3. They are sonorous.
Non-metals:
1. Non-metals are mostly soft solids that cannot be drawn into wires.
2. They are mostly gases and are not malleable.
3. They do not produce sound when struck.
APPEARS IN
संबंधित प्रश्न
Tick the most appropriate answer.
The constituents of a mixture are present in
- a fixed ratio
- a variable ratio,
- the ratio of 2: 1
- none of these
Correct the following statement:
CO and Co both represents cobalt.
Give Two Examples for Each of the Following: Non-metals
State the number of atoms of C6H12O6
Fill in the blank
A liquid non-metal is _______.
Differentiate between the terms –
elements, compounds & mixtures.
Select the correct answer from the choice given below.
The unreactive non-metallic element
Write the names of elements from the following symbol:
Br
Match the separation of components in List I with the most appropriate process in List II.
| List I | List II |
| 1. Naphthalene from naphthalene & sodium chloride. | A: Separating funnel |
| 2. Cream from milk. | B: Sublimation |
| 3. Kerosene oil from kerosene oil & water. | C: Boiling |
| 4. Lead nitrate from an aqueous solution of lead nitrate. | D: Centrifugation |
| 5. Ammonia from an aqueous solution of ammonia. | E: Distillation |
The diagram represents fractional distillation for the separation of mixtures. Answer the following :
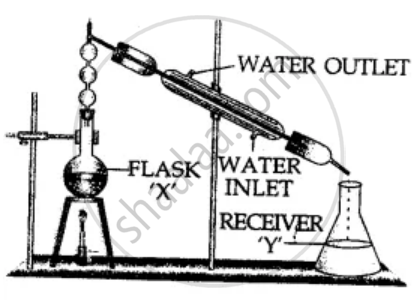
- Can two immiscible liquids be separated by this process?
- Separation of liquids by this process is based on which physical property?
- If methyl alcohol & water are to be separated, which liquid would remain in flask ‘X’ after condensation.
- Give a reason for the above answer.
- State the purpose of the fractionating column in the apparatus.
