Advertisements
Advertisements
प्रश्न
Give an example for a complex of the type [Ma2b2c2] where a, b, c are monodentate ligands and give the possible isomers.
Advertisements
उत्तर
The octahedral complexes of [Ma2b2c2] type can exist in five geometrical isomers.
The five geometrical isomers for the complex ion [PtCl2(NH3)2(py)2]2+are shown below.

(I)
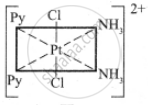
(II)
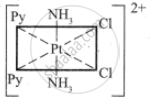
(III)
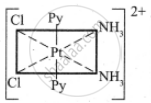
(IV)
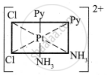
(V)
APPEARS IN
संबंधित प्रश्न
The sum of primary valence and secondary valence of the metal M in the complex [M(en)2(Ox)]Cl is ____________.
A complex has a molecular formula \[\ce{MSO4Cl.6H2O}\] The aqueous solution of it gives white precipitate with Barium chloride solution and no precipitate is obtained when it is treated with silver nitrate solution. If the secondary valence of the metal is six, which one of the following correctly represents the complex?
As per IUPAC guidelines, the name of the complex [Co(en)2(ONO)Cl]Cl is
Choose the correct statement.
Arrange the following in order of increasing molar conductivity.
i) \[\ce{Mg[Cr(NH3) (Cl)5]}\]
ii) \[\ce{[Cr(NH3)5 Cl]3 [CoF6]2}\]
iii) \[\ce{[Cr(NH3)3 Cl3]}\]
Give one test to differentiate \[\ce{[Co(NH3)5Cl]SO4}\] and \[\ce{[Co(NH3)5SO4]Cl}\].
In an octahedral crystal field, draw the figure to show the splitting of d orbitals.
What is crystal field stabilization energy (CFSE)?
Discuss briefly the nature of bonding in metal carbonyls.
What are the limitations of VB theory?
