Advertisements
Advertisements
प्रश्न
Explain why, osmosis can be considered to be a special kind of diffusion. Classify the following into
(i) osmosis, and (ii) diffusion :
- swelling up of a raisin on keeping in water
- spreading of virus on sneezing
- earthworm dying on coming in contact with common salt
- shrinking of grapes kept in thick sugar syrup
- preserving of pickles in salt
- spreading of smell of cake being baked in the kitchen
- aquatic animals using oxygen dissolved in water during respiration
Advertisements
उत्तर
Osmosis is considered to be a special kind of diffusion because it takes place only through a semi-permeable membrane, which allows only solvent particles to pass through it.
- Osmosis: a, c, d and e
- Diffusion: b, f and g.
APPEARS IN
संबंधित प्रश्न
Fill in the blank
All matter is made up of tiny particles called ______
Which type of elements, metals or non-metals, show the property of brittleness ?
Fill in the following blanks with suitable words :
Brass is an alloy which is considered a ..............
State any two properties for believing that aluminium is a metal.
Give reason why graphite is used for making elecrode in a dry cell.
Choose one term from the following which includes the other three :
aerosol, emulsion, colloid, sol
What will happen if a saturated solution is :
- heated
- cooled
What is the difference between colloids and suspensions ?
When the solid A is added to water, it dissolves with the evolution of a lot of heat and making little explosions to form two products B and C. The properties of products B and C are entirely different from those of solid A as well as water. Moreover, products B and C cannot be reconverted into solid A and water. When another solid D is added to water, it dissolves with the absorption of a little heat to form a product E which cools down. The product E shows the properties of both, solid D as well as water. Moreover, product E can be converted into solid D and water.
(a) What type of change occurs when solid A is dissolved in water ? Why ?
(b) What type of change occurs when solid D is dissolved in water ? Why ?
(c) Name a metal which you think could behave like solid A. Also name the products B and C.
(d) Name the solid D if it is the one which is used in making ordinary dry cells.
(e) Name the process by which D can be recovered from E.
The solubility of ammonium chloride in water at various temperatures is given below :
| Temperature | : | 10°C | 20°C | 40°C | 60°C | 80°C |
| Solubility | : | 24 g | 37 g | 41 g | 55 g | 66 g |
What mass of ammonium chloride would be needed to make a saturated solution of ammonium chloride in fifty grams of water at 40°C ?
Name the process by which the various gases of the air are separated.
What is the general name of the process by which tea-leaves are separated from prepared tea ?
Name the process by which all the dye can be recovered from black ink.
Write a method to separate a mixture of sand and potash alum.
You are given a mixture of iodine in alcohol called tincture iodine. Which method will you use to recover both, iodine as well as alcohol, from this mixture ?
There are three liquids A, B and C, all having different densities and different boiling points. Liquids A and C are organic in nature whereas liquid B is considered to be inorganic. When liquids A and B are put together in a container, they form a single layer. On the other hand, when, liquids B and C are mixed, they form two separate layers :
(a) Which process will you use to separate a mixture of A and B ?
(b) Which method will you use to separate a mixture of B and C ?
(c) Name the liquids which would behave like (i) A (ii) B and (iii) C.
Justify your answer.
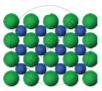
| Figure 1 | Figure 2 | Figure 3 |
 |
 |
 |
The arrangement of particles in three different phases of matter is shown above.
- Which state is represented by Fig. 1?
- In which state will the inter-particle attraction be maximum?
- Which one of them cannot be contained in an open vessel?
- Which one can take the shape of its container?
Complete the following table:
| S.No. | CELSIUS | KELVIN |
| 1. | 90°C | 363 K |
| 2. | ______ | 283 K |
| 3. | 63 °C | ______ |
| 4. | 250°C | ______ |
| 5. | ______ | 303 K |
Which statement is not correct for a mixture
Write a scientific reason.
Lemon sherbat has sweet, sour and salty taste and it can be poured in a glass.
