Advertisements
Advertisements
प्रश्न
Explain the thermodynamics of adiabatic process to obtain adiabatic constant for rigid diatomic molecule of a gas?
Advertisements
उत्तर
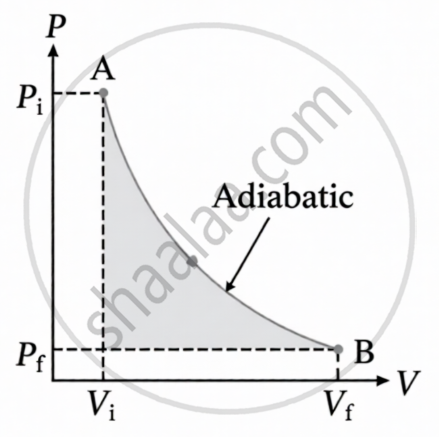
An adiabatic process is one in which no heat is transferred to or from the system. The P–V diagram of an adiabatic process is shown in the figure. In this instance, Q equals zero. The system is either completely insulated from the environment or the process proceeds rapidly to prevent any heat exchange. Δ U = −W for an adiabatic transformation.
W is positive when a system expands adiabatically due to the work done by the system, while Δ U is negative as a result of the system's internal energy decreasing.
W is negative when a system is compressed adiabatically, as work is performed on the system, and Δ U is positive. In an adiabatic process, the system's internal energy increases.
PVγ = C for an adiabatic process, where γ is the ratio of the specific heat at constant pressure to the specific heat at constant volume. γ is equivalent to `c_P/c_V`.
In the case of diatomic gases, the adiabatic ratio, also known as γ, is `7/5` for moderate temperature changes.
