Advertisements
Advertisements
प्रश्न
Explain the mechanism of alkaline hydrolysis of bromomethane.
स्पष्ट कीजिए
Advertisements
उत्तर
The reaction between methyl bromide and hydroxide ion to give methanol follows second-order kinetics; that is, the rate of this reaction depends on the concentration of two reacting species, namely, methyl bromide and hydroxide. Hence, it is called a substitution nucleophilic bimolecular SN2.
\[\ce{CH3Br + OH^\ominus -> CH3OH + Br^\ominus}\]
∴ Rate = \[\ce{k[CH3Br][OH^\ominus]}\]
The SN2 mechanism is a single-step mechanism with simultaneous bond breaking and bond forming.
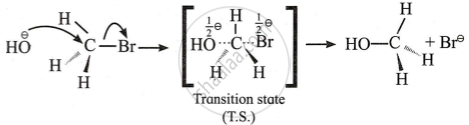
Approach of the nucleophile (Backside attack):
- The nucleophile \[\ce{(OH^\ominus)}\] slowly approaches the carbon atom from the opposite side of the C-Br bond.
- The C-OH weak bond is formed, while the existing C-Br bond gradually weakens.
- It is a slow process, and hence, it is the rate-determining step (R.D.S.).
Transition state:
- With the approach of \[\ce{(OH^\ominus)}\] group and the gradual departure of \[\ce{(Br^\ominus)}\], a stage comes where the central atom is attached to five substituents. This state is known as the transition state of the reaction.
- After this, \[\ce{(Br^\ominus)}\] leaves the compound and we get methanol.
- When an SN2 reaction is brought about at chiral carbon, the product is found to have the opposite configuration compared to that of the substrate. This is known as the inversion of configuration.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
2025-2026 (March) Official Question Paper
