Advertisements
Advertisements
प्रश्न
Explain the magnetic properties of [Ni(CN4)]2–.
Advertisements
उत्तर
Magnetic properties of [Ni(CN4)]2–:
- In [Ni(CN4)]2– ion, Ni2+ undergoes dsp2 hybridization.
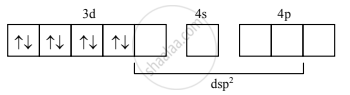
- Four vacant dsp2 hybrid orbitals of Ni2+ overlap with four orbitals of CN– ions to form Ni–CN coordinate bonds.
Configuration after the complex formation becomes:
- The complex has no unpaired electrons and hence, diamagnetic.
APPEARS IN
संबंधित प्रश्न
Choose the most correct option.
Which of the following complexes is chiral?
1. [Co(en)2Cl2]⊕
2. [Pt(en)Cl2]
3. [Cr(C2O4)3]3-
4. [Co(NH3)4Cl2]⊕
Answer in brief.
Classify the following complexes as homoleptic and heteroleptic [Cu(NH3)4]SO4, [Cu(en)2(H2O)Cl]2⊕, [Fe(H2O)5 (NCS)]2⊕, tetraammine zinc (II) nitrate.
Answer the following question.
What are cationic, anionic, and neutral complexes? Give one example of each.
Explain homoleptic and heteroleptic complexes.
Which of the following is CORRECT for [Co(NH3)5Br]Br2?
Identify the tetradentate ligand from the following.
Which statement from following is true for a complex hexaminecobalt (III) chloride?
Which from following complexes, the central metal ion does NOT obey BAN rule?
(Atomic number: Pt = 78, Cu = 29, Zn = 30, Fe = 26)
Which among the following complexes is NOT a heteroleptic complex?
Which among the following compounds is cationic complex?
Identify cationic complex from the following.
Which coordinate complex from the following has a complex ion with positive charge?
Explain cationic, anionic and neutral sphere complexes w ith example.
The correct order of ligands in the spectrochemical series is ______.
Which among the following cations forms a least stable complex if the ligand is same?
What is the number of donor atoms in dimethylglyoximato ligand?
Explain the classification of coordination complexes on the basis of charge on the complex.
Homoleptic complex from the following complexes is ______.
