Advertisements
Advertisements
प्रश्न
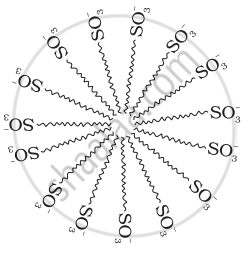
Draw the diagram showing micelle formation by the following detergent.
\[\ce{CH3(CH2)10CH2OS\overset{-}{O}3\overset{+}{N}a}\]
टिप्पणी लिखिए
Advertisements
उत्तर
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
