Advertisements
Advertisements
प्रश्न
Describe the structure of diborane.
Advertisements
उत्तर
In diborane, two BH2 units are linked by two bridged hydrogens. Therefore, it has eight B-H bonds. However, diborane has only 12 valance electrons and are not sufficient to form normal covalent bonds. The four-terminal B-H bonds are normal covalent bonds (two centre – two-electron bond or 2c-2e bond). The remaining four electrons have to used for the bridged bonds, i.e. two-three centred B-H-B bonds utilise two electrons each.
Hence, these bonds are three centre – two-electron bonds. The bridging hydrogen atoms are in a plane as shown in the figure. In dibome, the boron is sp3 hybridised. Three of the four sp3 hybridised orbitals contains a single electron and the fourth orbital is empty.
Two of the half-filled hybridised orbitals of each boron overlap with the two hydrogens to form four-terminal 2c-2e bonds, leaving one empty and one half filled hybridised orbitals on each boron. The Three centre – two-electron bonds, B-H-B bond formation involves overlapping the half-filled hybridised orbital of one boron, the empty hybridised orbital of the other boron and the half-filled 1s orbital of hydrogen.
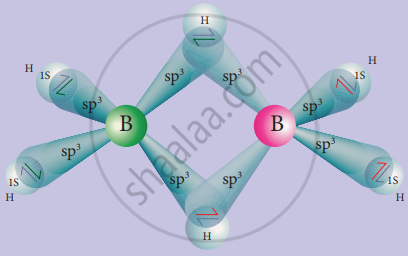
Structure of diborane
