Advertisements
Advertisements
प्रश्न
Describe simple experiment to show that particles of matter are closely packed in solids and less in liquids.
Advertisements
उत्तर
Experiment – To show that particles are closely packed in solids and less in liquids
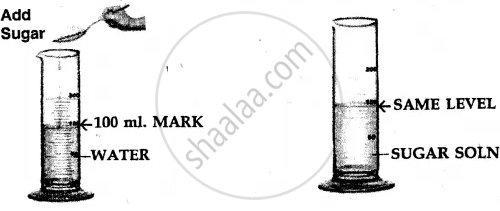
Procedure –
- Take a measuring cylinder and add 100 ml. of water to it.
- Add crystals of sugar [solid] to the water and stir carefully to obtain a sugar solution.
- The volume of water after the addition of sugar remains the
same i.e. at 100 ml. mark.
Conclusion –
- Particles of a liquid [e.g. water] are less closely packed.
- Particles of a solid [e.g. sugar] are closely packed.
- The sugar particles fill the intermolecular spaces, which must be existing between the water molecules, and hence the level of water in the measuring cylinder does not rise.
APPEARS IN
संबंधित प्रश्न
Fill in the blank:
The process in which a solid directly changes into a gas is called ....................
Why can a piece of chalk be broken easily into smaller pieces while a coal piece cannot be broken easily?
What are the two conditions for the interconversion of states of matter ?
Fill in the blank:
All matter is made up of tiny particles called..................
Write whether the following statement is true or false.
All solids, on heating, first change to the liquid and then to the gaseous state always.
A glass beaker is half-filled with water and an empty glass tumbler is inverted & lowered inside the glass beaker. State your observations on tilting the tumbler below the level of the water in the glass beaker. Give a reason for your answer.
State the term which refers to the conversion of a substance on heating from.
- solid-state to liquid state
- liquid state to vapour state
- vapour state to liquid state
- liquid state to solid-state.
State in which of the following examples ie. a piece of wood, water, light gas is the Inter-particle space maximum.
State in which of the following examples ie. a piece of wood, water, light gas is the Inter-particle attraction maximum.
Complete the statement by filling the blank with the correct word given below.
In naphthalene, the inter-particle attraction is _______.
