Advertisements
Advertisements
प्रश्न
Answer the following question.
Why is the wave theory of electromagnetic radiation not able to explain the photoelectric effect? How does a photon picture resolve this problem?
Advertisements
उत्तर
There are three main drawbacks:
- Intensity: If we consider light as a wave then as the intensity of the light is increased the amplitude of the oscillation of the electron will increase. Thus, as the intensity of the incident light is increased the maximum kinetic energy of the emitted electron will also increase. But it was observed that the kinetic energy of the emitted electrons does not depend on the intensity whereas the magnitude of the photoelectric current increases with the frequency.
- Frequency: If we consider the light as a wave then the photoelectric emission should happen on any frequency, but it was observed that the electrons are emitted after a particular frequency. If the frequency of the incident light is lesser than this frequency there is no photoelectric emission observed.
- Time Delay: According to the wave theory the energy is uniformly distributed over the wavefront. As the light falls on the metallic surface, it will take some time for the electron to gain sufficient energy to get emitted. But experimentally it was observed that the electrons are emitted instantaneously as the light falls on the metallic surface.
How the photon theory can explain the photoelectric effect:
- According to photon theory increasing the intensity means increasing the number of photons that do not change the maximum kinetic energy but changes the number of ejected electrons.
- The energy of a photon is given as E = hf that explains the dependence of the energy on the frequency, after a particular frequency of a photon that is threshold frequency there is photoelectric emission.
- As soon as a photon falls on the metallic surface it is absorbed, hence the electron is ejected instantaneously. Hence, all these are in accordance with the experimental observations.
संबंधित प्रश्न
Is the formula you employ in (a) valid for calculating radius of the path of a 20 MeV electron beam? If not, in what way is it modified?
An electron gun with its collector at a potential of 100 V fires out electrons in a spherical bulb containing hydrogen gas at low pressure (∼10−2 mm of Hg). A magnetic field of 2.83 × 10−4 T curves the path of the electrons in a circular orbit of radius 12.0 cm. (The path can be viewed because the gas ions in the path focus the beam by attracting electrons, and emitting light by electron capture; this method is known as the ‘fine beam tube’ method. Determine e/m from the data.
What is so special about the combination e/m? Why do we not simply talk of e and m separately?
If light of wavelength 412.5 nm is incident on each of the metals given below, which ones will show photoelectric emission and why?
| Metal | Work Function (eV) |
| Na | 1.92 |
| K | 2.15 |
| Ca | 3.20 |
| Mo | 4.17 |
A light beam of wavelength 400 nm is incident on a metal plate of work function 2.2 eV. (a) A particular electron absorbs a photon and makes two collisions before coming out of the metal. Assuming that 10% of the extra energy is lost to the metal in each collision, find the kinetic energy of this electron as it comes out of the metal. (b) Under the same assumptions, find the maximum number of collisions the electron can suffer before it becomes unable to come out of the metal.
A horizontal cesium plate (φ = 1.9 eV) is moved vertically downward at a constant speed v in a room full of radiation of wavelength 250 nm and above. What should be the minimum value of v so that the vertically-upward component of velocity is non-positive for each photoelectron?
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
In the arrangement shown in the figure, y = 1.0 mm, d = 0.24 mm and D = 1.2 m. The work function of the material of the emitter is 2.2 eV. Find the stopping potential V needed to stop the photocurrent.
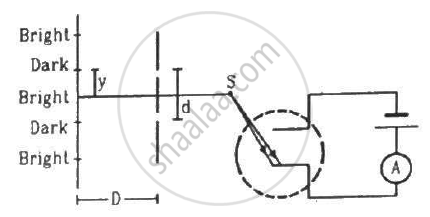
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
In various experiments on photo electricity, the stopping potential for a given frequency of the incident radiation is ______.
The electromagnetic theory of light failed to explain ______.
Cathode rays can be deflected by
