Advertisements
Advertisements
प्रश्न

(a) Name the experiment illustrate above.
(b) Which property of hydrogen chloride is demonstrated by this experiment?
(c) State the colour of the water that has entered the round-bottomed flask.
Advertisements
उत्तर
(a) Fountain experiment.
(b) This experiment demonstrates that hydrogen chloride is very soluble in water.
(c) Red
APPEARS IN
संबंधित प्रश्न
When dilute HCl is added to a salt Q, a brisk effervescence is produced and the gas turns lime water milky.
When NH4OH solution is added to the above mixture (after adding dilute HCl), it produces a white precipitate which is soluble in excess NH4OH solution.
The aim of the Fountain experiment is to prove that ______.
Study the figure given below and answer the questions which follow:
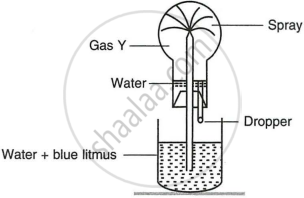
- Identify the gas Y.
- What property of gas Y does this experiment demonstrate?
- Name another gas which has the same property and can be demonstrated through this experiment.
Name the following:
Gas obtained by treating metals with hydrochloric acid.
Name the drying agent used to dry HCl gas.
Choose the correct answer from the options given below: HCl gas can be prepared by direct combination of hydrogen and chlorine gas in presence of
Name two gases which combine chemically to form a solid.
Give reason for the following:
Dilute hydrochloric acid cannot be concentrated by distilling (boiling) the dilute acid.
The following question is pertaining to the laboratory preparation of hydrogen chloride gas.
Write the equation for its preparation, mentioning the condition required.
What property of hydrogen chloride is demonstrated when it is collected by downward delivery (Upward displacement)?
