Advertisements
Advertisements
प्रश्न
A burning candle shows both physical and chemical changes. Explain ?
Burning of a candle is both a physical and chemical change.
Advertisements
उत्तर १
A burning candle produces wax vapours which solidify and form wax again is physical change. At the same time wax on burning produces water vapours and carbondioxide which escape into the atmosphere which are new substances with new properties. It is a chemical change.
Hence burning of candle shows both physical and chemcial changes.
उत्तर २
When a candle is lighted, some of the solid wax first melts and turns into liquid, then it turns into vapours to produce a flame. New substances CO2 and H2O vapours are formed along with the evolution of light and heat energy. This shows a chemical change. When some of the molten wax drops to the floor, it again solidifies. Which shows a physical change. Thus the melting of candle wax is a physical change and the production of CO2 and H2O represents chemical change.
संबंधित प्रश्न
Classify the following as chemical or physical change:
Burning of paper and wood
Fill in the blank.
A new substance is formed in a ............... change.
Write true or false for the statement
Decomposition of leaves is a fast change.
Write short answer.
Sublimation is classified as a physical change. Why ?
Is burning a physical change or a chemical change? Why?
State whether the following is physical or chemical changes.
heating a mixture of iron filings and sulphur—
Tick (√) the correct alternative from the choice given for the following statement.
A substance which can not sublime is
Name two reversible changes.
Solve this crossword by using the clues that follow-
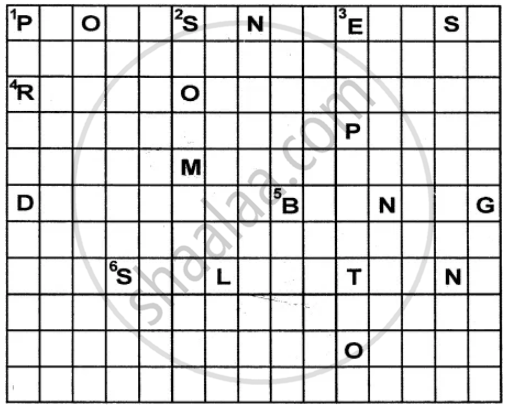
Across
1. Energy in the form of sunlight is absorbed by the green plants in this process.
4. The reddish-brown substance formed over iron in the presence of oxygen and moisture.
5. This change is permanent and irreversible.
6. The process in which a solid directly changes into gaseous state.
Down
1 . It is the change that takes place in case of swinging pendulum of a clock. –
2. Occurrence of this is a non-periodic change as well as a natural change.
3. It is a physical change.
State one instance where water undergoes a physical change and one in which it undergoes a chemical change.
Classify the following as physical or chemical change :
Formation of curd from milk
Classify the following as a physical or a chemical change.
Drying of wet clothes
What will you take into account while identifying the following?
A chemical change in a substance.
Photosynthesis is a natural process by which green plants manufacture food in the presence of sunlight.
State the conclusions which can be drawn, to represent photosynthesis as a physical or a chemical change.
Give a reason why – Heating a platinum wire is a reversible change but, heating a magnesium wire is an irreversible change.
Select the correct answer from the choice given below to complete the sentence
The dissolution of lead nitrate in water is deemed as a _________ change.
Give the reason why the following is considered as chemical changes.
A piece of magnesium strip is dropped into a beaker containing dilute hydrochloric acid.
When ice melts to form water, change occurs in its
Physical Change: Boiling::Chemical Change: ______.
What kind of change is shown by tearing of paper?
