Advertisements
Advertisements
Reduction potential of two metals M1 and M2 are \[\ce{E^0_{{M_1^{2+}|M_1}}}\] = −2.3 V and \[\ce{E^0_{{M_2^{2+}|M_2}}}\] = 0.2 V. Predict which one is better for coating the surface of iron.
Given: \[\ce{E^0_{{Fe^{2+}|Fe}}}\] = −0.44 V
Concept: undefined >> undefined
A copper electrode is dipped in 0.1 M copper sulphate solution at 25°C. Calculate the electrode potential of copper.
[Given: \[\ce{E^0_{{Cu^{2+}|Cu}}}\] = 0.34 V]
Concept: undefined >> undefined
Advertisements
For the cell \[\ce{Mg_{(s)}|Mg^{2+}_{( aq)}||Ag^+_{( aq)}|Ag_{(s)}}\], calculate the equilibrium constant at 25°C and maximum work that can be obtained during operation of cell.
Given: \[\ce{E^0_{{Mg^{2+}|Mg}}}\] = −2.37 V and \[\ce{E^0_{{Ag^{+}|Ag}}}\] = 0.80 V
Concept: undefined >> undefined
Write a note on sacrificial protection.
Concept: undefined >> undefined
An alkene “A” on reaction with O3 and Zn – H2O gives propanone and ethanol in equimolar ratio. Addition of HCl to alkene “A” gives “B” as the major product. The structure of product “B” is
Concept: undefined >> undefined
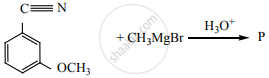
Product ‘P’ in the above reaction is:
Concept: undefined >> undefined
Ammonium salt of benzoic acid is heated strongly with P2O5 and the product so formed is reduced and then treated with NaNO2/HCl at low temperature. The final compound formed is ____________.
Concept: undefined >> undefined
Identify X in the sequence given below
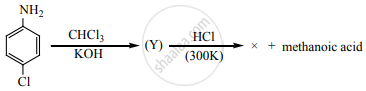
Concept: undefined >> undefined
The major product of the following reaction:
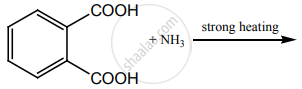
Concept: undefined >> undefined
Write a short note on the following.
Coupling reaction
Concept: undefined >> undefined
Write a short note on the following.
Gomberg reaction
Concept: undefined >> undefined
Account for the following.
Diazonium salts of aromatic amines are more stable than those of aliphatic amines.
Concept: undefined >> undefined
Account for the following.
Amines are more basic than amides.
Concept: undefined >> undefined
Identify A, B, C and D.
\[\ce{aniline + benzaldehyde -> A ->[Conc. HNO3][B] C + D}\]
Concept: undefined >> undefined
A dibromo derivative (A) on treatment with KCN followed by acid hydrolysis and heating gives a monobasic acid (B) along with the liberation of CO2. (B) on heating with liquid ammonia followed by treating with Br2/KOH gives (C) which on treating with NaNO2 and HCl at low temperature followed by oxidation gives a monobasic acid (D) having molecular mass 74. Identify A to D.
Concept: undefined >> undefined
What are the functions of lipids in living organisms?
Concept: undefined >> undefined
Write a note on synthetic detergents.
Concept: undefined >> undefined
Explain the mechanism of cleansing action of soaps and detergents.
Concept: undefined >> undefined
Identify the product (s) is / are formed when 1 – methoxy propane is heated with excess HI. Name the mechanism involved in the reaction
Concept: undefined >> undefined
Write the Zwitter ion structure of alanine
Concept: undefined >> undefined
