Please select a subject first
Advertisements
Advertisements
Consider the oxides Li2O, CO2, B2O3.
Which oxide would you expect to be the most basic?
Concept: undefined >> undefined
Consider the oxides Li2O, CO2, B2O3.
Which oxide would be the most acidic?
Concept: undefined >> undefined
Advertisements
Consider the oxides Li2O, CO2, B2O3.
Give the formula of an amphoteric oxide.
Concept: undefined >> undefined
Choose the correct option.
Thallium shows different oxidation state because __________
Concept: undefined >> undefined
Identify the group 14 element that best fits the following description.
Nonmetallic element
Concept: undefined >> undefined
Identify the group 14 element that best fits the following description.
Form the most acidic oxide
Concept: undefined >> undefined
Identify the group 14 element that best fits the following description.
They prefer the +2 oxidation state.
Concept: undefined >> undefined
Identify the group 14 element that best fits the following description.
Forms strong π bonds.
Concept: undefined >> undefined
Give reason.
Ga3+ salts are better reducing agents while Tl3+ salts are the better oxidizing agent.
Concept: undefined >> undefined
Give reason.
PbCl4 is less stable than PbCl2.
Concept: undefined >> undefined
Give the formula of a compound in which carbon exhibit an oxidation state of +4
Concept: undefined >> undefined
Give the formula of a compound in which carbon exhibit an oxidation state of +2
Concept: undefined >> undefined
Give the formula of a compound in which carbon exhibit an oxidation state of −4
Concept: undefined >> undefined
Explain the trend in the oxidation state of elements from nitrogen to bismuth.
Concept: undefined >> undefined
State true or false. Correct the false statement.
The acidic nature of oxides of group 13 increases down the graph.
Concept: undefined >> undefined
Which of the following will exhibit geometrical isomerism?
Concept: undefined >> undefined
Complete the following reaction sequence:
\[\ce{HC ≡ CH ->[LiNH2] A ->[CH3CH2Cl] B ->[HBr] C}\]
Comment on the acidic nature of B.
Concept: undefined >> undefined
Explain the following:
Turmeric powder can be used as an antiseptic.
Concept: undefined >> undefined
Identify the functional group in the following molecule:
Aspirin
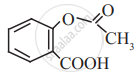
Concept: undefined >> undefined
Identify the functional group in the following molecule:
Paracetamol

Concept: undefined >> undefined
