Please select a subject first
Advertisements
Advertisements
Write the reaction to convert propene to isopropyl alcohol.
Concept: undefined >> undefined
What is the action of following on but-2-ene?
dil. alkaline KMnO4
Concept: undefined >> undefined
Advertisements
Write bond line formula and condensed formula for the following compound.
2, 2, 4, 4-tetramethylpentane
Concept: undefined >> undefined
Write bond line formulae and condensed formulae for the following compound.
2, 2, 4, 4- tetramethylpentane
Concept: undefined >> undefined
Write the structural formula for the following name and also write correct IUPAC name for it.
2,2,3-trimethylpentan-4-ol
Concept: undefined >> undefined
Write the structural formulae for the following name and also write correct IUPAC name for it.
2, 2, 3-trimethylpentan-4-ol
Concept: undefined >> undefined
Write dash formulae for the following bond line formulae.
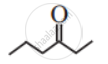
Concept: undefined >> undefined
What is the action of following on but-2-ene ?
dil. alkaline \[\ce{KMnO4}\]
Concept: undefined >> undefined
Write dash formula for the following bond line formula.
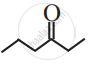
Concept: undefined >> undefined
What is the action of following on but-2-ene?
dil. alkaline \[\ce{KMnO4}\]
Concept: undefined >> undefined
Write the reaction to convert propene to isopropyl alcohol.
Concept: undefined >> undefined
What is the action of following on but-2-ene ?
dil. alkaline \[\ce{KMnO4}\]
Concept: undefined >> undefined
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\ce{H\phantom{...}H\phantom{...}H\phantom{...}H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - C - H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H\phantom{...}H\phantom{...}H\phantom{...}H}
\end{array}\]
Concept: undefined >> undefined
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\\
\ce{N ≡ C - C - C - C ≡ N}\\
|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}
\end{array}\]
Concept: undefined >> undefined
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\phantom{.........}\ce{H\phantom{...}H\phantom{...}H}\phantom{......}\ce{O}\phantom{.}\\
\phantom{.........}|\phantom{....}|\phantom{....}|\phantom{.....}//\phantom{..}\\
\ce{H - C - C - C - C}\\
\phantom{...........}|\phantom{....}|\phantom{....}|\phantom{.....}\backslash\phantom{.....}\\
\phantom{..........}\ce{H\phantom{...}H\phantom{...}H}\phantom{.....}\ce{OH}\phantom{..}
\end{array}\]
Concept: undefined >> undefined
Write dash formulae for the following bond line formulae.
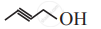
Concept: undefined >> undefined
Acetone and acetaldehyde are the ozonolysis products of an alkene. Write the structural formula of an alkene and give IUPAC name of it.
Concept: undefined >> undefined
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{...............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH_2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
Concept: undefined >> undefined
Write bond line formulae and condensed formulae for the following compound.
2, 2, 4, 4-tetramethylpentane
Concept: undefined >> undefined
Distinguish between the meaning of the terms adsorption and absorption. Give one example of.
Concept: undefined >> undefined
