Please select a subject first
Advertisements
Advertisements
Which among the following is a globular protein?
Concept: undefined >> undefined
Aniline on reaction with Bromine water at room temperature gives ____________.
Concept: undefined >> undefined
Advertisements
When propanamide is treated with bromine and aqueous sodium hydroxide, the compound formed is ____________.
Concept: undefined >> undefined
If two moles of an ideal gas at 546 K occupy a volume of 44.8 L. What is the pressure of ideal gas at 546 K? (R = 0.0821 L atm mol-1 K-1)
Concept: undefined >> undefined
Identify the side chain –R present in amino acid lysine.
Concept: undefined >> undefined
Which among the following is basic amino acid?
Concept: undefined >> undefined
Which of the following functional groups is reduced by diborane?
Concept: undefined >> undefined
Which of the following compounds is obtained when ethanoic anhydride is treated with water?
Concept: undefined >> undefined
What is the position of elements La (Z = 57) and Ce (Z = 58) respectively in the long form of periodic table?
Concept: undefined >> undefined
Which of the following elements belongs to first inner transition series?
Concept: undefined >> undefined
Which of the following polymers is used to obtain adhesives?
Concept: undefined >> undefined
Which of the following carboxylic acids ha highest acidic strength?
Concept: undefined >> undefined
When dry cell is in use the change taking place at cathode is ______.
Concept: undefined >> undefined
Which among the following elements belongs to second inner transition series?
Concept: undefined >> undefined
Which among the following elements does NOT form oxocation?
Concept: undefined >> undefined
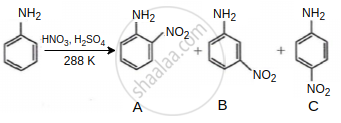
Consider the given reaction, percentage yield of:
Concept: undefined >> undefined
Which oxide of nitrogen is obtained on heating ammonium nitrate at 250°C?
Concept: undefined >> undefined
In Duma's method, 0.52 g of an organic compound on combustion gave 68.6 mL N2 at 27°C and 756 mm pressure. What is the percentage of nitrogen in the compound?
Concept: undefined >> undefined
What is the general molecular formula of the products obtained on heating lanthanoids (Ln) with sulphur?
Concept: undefined >> undefined
100 g of an ideal gas is kept in a cylinder of 416 L volume at 27°C under 1.5 bar pressure. The molar mass of the gas is ______ g mol−1.
Concept: undefined >> undefined
