Please select a subject first
Advertisements
Advertisements
Identify the coordinate complex having ambidentate ligand from following.
Concept: undefined >> undefined
Which of the following molecule does not obey octet rule?
Concept: undefined >> undefined
Advertisements
How many carbon atoms are present in a molecule of benzyl methyl ketone?
Concept: undefined >> undefined
In which of the following compound intra molecular hydrogen bonding is present?
Concept: undefined >> undefined
If 'Q' is the magnitude of charge and 'r' is the distance between the centres of positive and negative charges then dipole moment (µ) is given by ______.
Concept: undefined >> undefined
Identify the type of intermolecular force present between benzene and ammonia.
Concept: undefined >> undefined
Which of the following is an example of physisorption?
Concept: undefined >> undefined
What is the molar mass of a compound represented below?
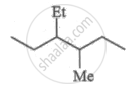
Concept: undefined >> undefined
Which among the following compounds has highest boiling point?
Concept: undefined >> undefined
Which of the following alcohols is NOT having \[\ce{C_{sp^3} - OH}\] bond?
Concept: undefined >> undefined
What is the average rate of reaction when the change in concentration of product is 0.05 Min 20 seconds?
Concept: undefined >> undefined
Extent of physisorption of a gas increases with ______.
Concept: undefined >> undefined
Which of the following compound when treated with dibenzyl cadmium yields benzyl methyl ketone?
Concept: undefined >> undefined
Which of the following carboxylic acids is a tricarboxylic acid?
Concept: undefined >> undefined
Average rate of reaction for the following reaction.
\[\ce{2SO2 (g) + O2(g) -> 2SO3 (g)}\] is written as ______.
Concept: undefined >> undefined
The electric cookers have a coating that protects them against fire. The coating is made of ______.
Concept: undefined >> undefined
Which among the following carboxylic acids is strongest acid?
Concept: undefined >> undefined
What is the number of \[\ce{-COOH}\] groups present in Phthalic acid?
Concept: undefined >> undefined
For the reaction,
\[\ce{2NO(g) + O2(g) -> 2NO2(g) ->[d(NO2)][dt] = 0.052 mol dm^{-3} s^{-1}}\]
Calculate rate of consumption of NO (g).
Concept: undefined >> undefined
