Advertisements
Advertisements
Give the structural formula of the following organic compound:
Methanal
Concept: undefined >> undefined
Give the structural formula of the following organic compound:
But-2-yne
Concept: undefined >> undefined
Advertisements
Identify the cation in the following case:
Sodium hydroxide solution when added to Solution C gives a white precipitate which is insoluble in excess of sodium hydroxide solution.
Concept: undefined >> undefined
The Empirical formula of an organic compound is CHCl2.
If its relative molecular mass is 168, what is its molecular formula?
[At. Wt. C = 12, H = 1, Cl = 35.5]
Concept: undefined >> undefined
State one relevant observation for the following:
To the copper nitrate solution, initially, few drops of sodium hydroxide solution is added and then added in excess.
Concept: undefined >> undefined
Calculate:
Write the empirical formula of C8H18.
Concept: undefined >> undefined
Explain the following:
\[\ce{H3PO3}\] is not a tribasic acid.
Concept: undefined >> undefined
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
Concept: undefined >> undefined
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.
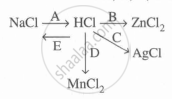
Concept: undefined >> undefined
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
Concept: undefined >> undefined
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
Concept: undefined >> undefined
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
Concept: undefined >> undefined
The metal nitrate which on thermal decomposition forms a black residue is ______.
Concept: undefined >> undefined
Give the IUPAC name of the organic compound represented by the structural formula given below:
\[\begin{array}{cc}
\ce{H}\phantom{...}\ce{Cl}\phantom{..}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - C - C - H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{Cl}\phantom{..}\ce{H}\phantom{...}\ce{H}\end{array}\]
Concept: undefined >> undefined
Give the IUPAC name of the organic compound represented by the structural formula given below:
\[\begin{array}{cc}
\phantom{.}\ce{H\phantom{...}H\phantom{...}O}\phantom{..}\\
\phantom{.}|\phantom{....}|\phantom{....}||\phantom{..}\\
\ce{H - C - C - C - OH}\\
\phantom{}|\phantom{....}|\phantom{......}\\
\phantom{}\ce{H\phantom{...}H}\phantom{......}
\end{array}\]
Concept: undefined >> undefined
The compound which does not have a double bond in its structure.
Concept: undefined >> undefined
The compound in its pure form turns into an ice like solid on cooling.
Concept: undefined >> undefined
The compound which is used for artificial ripening of fruits.
Concept: undefined >> undefined
Identify the gas evolved in the following reaction:
Copper carbonate is heated.
Concept: undefined >> undefined
Give a balanced equation for the following reaction:
Copper reacts with concentrated nitric acid.
Concept: undefined >> undefined
