Advertisements
Advertisements
In the manufacture of sulphuric acid by contact process Tyndall box is used to ____________.
Concept: undefined >> undefined
To get n-type of semiconductor, germanium should be doped with ____________.
Concept: undefined >> undefined
Advertisements
p-type semiconductors are formed when Si or Ge are doped with ____________.
Concept: undefined >> undefined
Hot conc. \[\ce{H2SO4}\] acts as the moderately strong oxidising agent. It oxidises both metals and non-metals. Which of the following element is oxidised by conc. \[\ce{H2SO4}\] into two gaseous products?
Concept: undefined >> undefined
Which of the following oxides behaves as conductor or insulator depending upon temperature?
Concept: undefined >> undefined
Which of the following is true about the charge acquired by p-type semiconductors?
Concept: undefined >> undefined
To get a n-type semiconductor from silicon, it should be doped with a substance with valence ______.
Concept: undefined >> undefined
Under the influence of electric field, which of the following statements is true about the movement of electrons and holes in a p-type semiconductor?
(i) Electron will move towards the positvely charged plate through electron holes.
(ii) Holes will appear to be moving towards the negatively charged plate.
(iii) Both electrons and holes appear to move towards the positively charged plate.
(iv) Movement of electrons is not related to the movement of holes.
Concept: undefined >> undefined
A perfect crystal of silicon (Figure) is doped with some elements as given in the options. Which of these options show n-type semiconductors?




Concept: undefined >> undefined
Explain why does conductivity of germanium crystals increase on doping with galium.
Concept: undefined >> undefined
How does the doping increase the conductivity of semiconductors?
Concept: undefined >> undefined
Match the types of colloidal systems given in Column I with the name given in Column II.
| Column I | Column II |
| (i) Solid in liquid | (a) Foam |
| (ii) Liquid in solid | (b) Sol |
| (iii) Liquid in liquid | (c) Gel |
| (iv) Gas in liquid | (d) Emulsion |
Concept: undefined >> undefined
What is the role of adsorption in froth floatation process used especially for concentration of sulphide ores?
Concept: undefined >> undefined
Which of the following statements are correct?
(i) S – S bond is present in \[\ce{H2S2O6}\].
(ii) In peroxosulphuric acid \[\ce{(H2SO5)}\] sulphur is in +6 oxidation state.
(iii) Iron powder along with \[\ce{Al2O3}\] and \[\ce{K2O}\] is used as a catalyst in the preparation of \[\ce{NH3}\] by Haber’s process.
(iv) Change in enthalpy is positive for the preparation of \[\ce{SO3}\] by catalytic oxidation of \[\ce{SO2}\].
Concept: undefined >> undefined
Complete the following reaction sequence.
\[\begin{array}{cc}
\ce{O}\phantom{...............................................}\\
||\phantom{...............................................}\\
\ce{CH3 - C - CH3 ->[(i) CH3MgBr][H2O] (A) ->[Na metal][Ether] (B) ->[CH3 - Br] (C)}
\end{array}\]
Concept: undefined >> undefined
Assertion: Formaldehyde is a planar molecule.
Reason: It contains sp2 hybridised carbon atom.
Concept: undefined >> undefined
Which of the following compound will not undergo azo coupling reaction with benzene diazonium chloride?
Concept: undefined >> undefined
The product of the following reaction is:

(i)
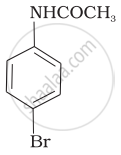
(ii)
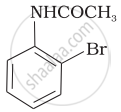
(iii)
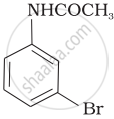
(iv)
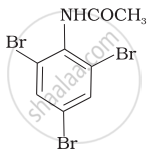
Concept: undefined >> undefined
Which of the following reactions belong to electrophilic aromatic substitution?
(i) Bromination of acetanilide
(ii) Coupling reaction of aryldiazonium salts
(iii) Diazotisation of aniline
(iv) Acylation of aniline
Concept: undefined >> undefined
Under what reaction conditions (acidic/basic), the coupling reaction of aryldiazonium chloride with aniline is carried out?
Concept: undefined >> undefined
