Advertisements
Advertisements
Type of stoichiometric defect shown by ZnS is ____________.
Concept: undefined >> undefined
Type of stoichiometric defect shown by AgBr is ____________.
Concept: undefined >> undefined
Advertisements
Which of the following is the most likely structure of CrCl3.6H2O if `1/3` of total chlorine of the compound is precipitated by adding AgNO3 to its aqueous solution?
Concept: undefined >> undefined
Which of the following complex will give a white precipitate with barium chloride solution?
Concept: undefined >> undefined
Schottky defect defines imperfection in the lattice structure of ____________.
Concept: undefined >> undefined
Which is not correct about the Schottky defects?
Concept: undefined >> undefined
Assertion: No compound has both Schottky and Frenkel defects.
Reason: Both defects change the density of the solid.
Concept: undefined >> undefined
Assertion: Due to Frenkel defect, there is no effect on the density of the crystalline solid.
Reason: In Frenkel defect, no cation or anion leaves the crystal.
Concept: undefined >> undefined
0.450 g of urea (mol. wt. 60) in 22.5 g of water show 0.170°C of elevation in boiling point. The molal elevation constant of water is:
Concept: undefined >> undefined
Exactly 1 g of urea dissolved in 75 g of water gives a solution that boils at 100.114°C at 760 torrs. The molecular weight of urea is 60.1. The boiling point elevation constant for water is:
Concept: undefined >> undefined
Most acidic oxide among the following is:
Concept: undefined >> undefined
Iodide of Millon's base is ____________.
Concept: undefined >> undefined
In the following sequence of the reactions, what is D?
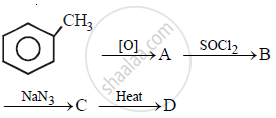
Concept: undefined >> undefined
The pesticide DDT slowly changes to __________.
Concept: undefined >> undefined
Phosgene is a common name for ____________.
Concept: undefined >> undefined
In which of the following conversions, phosphorus pentachloride is used as the reagent?
Concept: undefined >> undefined
Chloroform and conc. HNO3 reacts to produce ____________.
Concept: undefined >> undefined
B.H.C. is used as an ____________.
Concept: undefined >> undefined