Advertisements
Advertisements
Answer the following question.
What is observed when 2 mL of dilute hydrochloric acid is added to 1 g of sodium carbonate taken in a clean and dry test tube? Write a chemical equation for the reaction involved.
Concept: undefined >> undefined
| Human body is made up of five important components, of which water is the main component. Food, as well as potable water, are essential for every human being. The food is obtained from plants through agriculture. Pesticides are being used extensively for a high yield in the fields. These pesticides are absorbed by the plants from the soil along with water and minerals and from the water bodies these pesticides are taken up by the aquatic animals and plants. As these chemicals are not biodegradables, they get accumulated progressively at each trophic level. The maximum concentration of these chemicals gets accumulated in our bodies and greatly affects the health of our mind and body. |
- Why is the maximum concentration of pesticides found in human beings? (1)
- Give one method which could be applied to reduce our intake of pesticides through food to some extent. (1)
- Various steps in a food chain represent: (1)
- Food web
- Trophic level
- Ecosystem
- Biomagnification
- With regard to various food chains operating in an ecosystem, man is a: (1)
- Consumer
- Producer
- Producer and consumer
- Producer and decomposer
Concept: undefined >> undefined
Advertisements
1 g of copper powder was taken in a China dish and heated. What change takes place on heating? When hydrogen gas is passed over this heated substance, a visible change is seen in it. Give the chemical equations of reactions, the name and the color of the products formed in each case.
Concept: undefined >> undefined
What is thermit process?
Concept: undefined >> undefined
Name the substance that is getting oxidised in the thermit process.
Concept: undefined >> undefined
Write the name and molecular formula of a carbon compound having its name suffixed with "-ol" and having two carbon atoms in its molecule. With the help of a chemical equation indicate what happens when this compound is heated with excess conc. H2SO4.
Concept: undefined >> undefined
When a potassium iodide solution is added to a solution of lead (II) nitrate in a test tube, a precipitate is formed.
What is the colour of this precipitate? Name the compound precipitated.
Concept: undefined >> undefined
When a potassium iodide solution is added to a solution of lead (II) nitrate in a test tube, a precipitate is formed.
Write the balanced chemical equation for this reaction.
Concept: undefined >> undefined
What are decomposers? What will be the consequence if the decomposers are completely eradicated from an ecosystem? Give justification in support of your answer.
Concept: undefined >> undefined
A student mixes sodium sulphate powder in barium chloride powder. What change would the student observe on mixing the two powders? Justify your answer and explain how he can obtain the desired change.
Concept: undefined >> undefined
List two differences between the properties exhibited by covalent compounds and ionic compounds.
Concept: undefined >> undefined
Which of the following is the purest form of carbon?
Concept: undefined >> undefined
Which of the following ray diagrams is correct for the ray of light incident on a lens shown in figure?

 |
 |
| A | B |
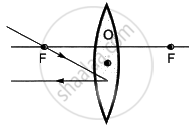 |
 |
| C | D |
Concept: undefined >> undefined
Which position of the object will produce a magnified virtual image, if a concave mirror of focal length 15 cm is being used?
Concept: undefined >> undefined
A ray of light is travelling in a direction perpendicular to the boundary of a parallel glass slab. The ray of light ______.
Concept: undefined >> undefined
Reema took 5ml of Lead Nitrate solution in a beaker and added approximately 4ml of Potassium Iodide solution to it. What would she observe?
Concept: undefined >> undefined
When ethyl alcohol and acetic acid are mixed, the resulting ester has a chemical formula ______.
Concept: undefined >> undefined
The correct structural formula of butanoic acid is -
Concept: undefined >> undefined
Acetic acid was added to a solid X kept in a test tube. A colourless, odourless gas Y was evolved. The gas was passed through lime water, which turned milky. It was concluded that ______.
Concept: undefined >> undefined
Carbon forms four covalent bonds by sharing its four valence electrons with four univalent atoms, e.g., hydrogen. After the formation of four bonds, carbon attains the electronic configuration of ______.
Concept: undefined >> undefined
